The pressure $P$ of an ideal diatomic gas varies with its absolute temperature $T$ as shown in figure. The molar heat capacity of gas during this process is ........... $R$ [$R$ is gas constant]
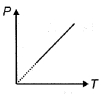

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
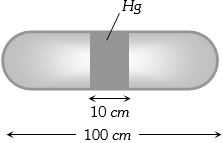
- 1A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 2An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :View Solution
- 3View SolutionA sample contains mixture of helium and oxygen gas. The ratio of root mean square speed of helium and oxygen in the sample, is:
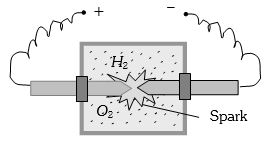
- 4A vessel contains $14 \,gm \,(7 $ moles) of hydrogen and $96\, gm$ ($9$ moles) of oxygen at $STP.$ Chemical reaction is induced by passing electric spark in the vessel till one of the gases is consumed. The temperature is brought back to it's starting value $273 K.$ The pressure in the vessel is ...... $atm$View Solution
- 5View SolutionIf the pressure in a closed vessel is reduced by drawing out some gas, the mean free path of the molecules
- 6What will be the average value of energy along one degree of freedom for an ideal gas in thermal equilibrium at a temperature $T \,?\left( k _{ B }\right.$ is Boltzmann constant)View Solution
- 7The ratio of specific heats $(\gamma )$ of a ideal gas is given byView Solution
- 8One mole of a monoatomic gas is mixed with three moles of a diatomic gas. The molecular specific heat of mixture at constant volume is $\frac{\alpha^{2}}{4} R J / mol\,K$; then the value of $\alpha$ will be $.......$ (Assume that the given diatomic gas has no vibrational mode.)View Solution
- 9A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
- 10A gas mixture consists of molecules of type $1, 2$ and $3$, with molar masses ${m_1} > {m_2} > {m_3}.$ ${V_{rms}}$ and $\overline K $ are the $r.m.s.$ speed and average kinetic energy of the gases. Which of the following is trueView Solution
