$\Delta W=20 \mathrm{KJ}$
$\Delta Q=16 \mathrm{KJ}$
$\therefore \Delta U=\Delta Q-\Delta W=-4 K J$
since, $U$ is a state function. Therefore, value of $DU$ in both expansions remain same.
Thus, for second expansion $:-$
$\Delta U=-4 K J, \Delta Q=9 K J$
$\therefore$ By first law of the thermodynamics
$\Delta \mathrm{W}=\Delta \mathrm{Q}-\Delta \mathrm{U}=13 \mathrm{KJ}$
Download our appand get started for free
Similar Questions
- 1If a Carnot’s engine functions at source temperature $127^o C$ and at sink temperature $87^o C$, what is its efficiency ....... $\%$View Solution
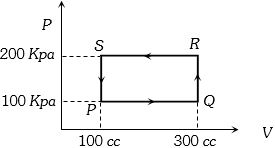
- 2A thermodynamic system is taken through the cycle $PQRSP$ process. The net work done by the system is ....... $J$View Solution
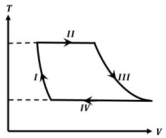
- 3One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
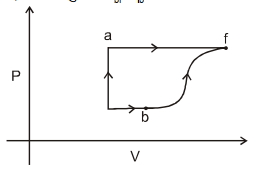
- 4A thermodynamic system is taken form an initial state $i$ with internal energy $U_1=100 \ J$ to the final state along two different paths iaf and ibf, as schematically shown in the fire. The work done by the system along the paths $af$, ib and bf are $W _{ af }=200 \ J , W _{ ID }=50 \ J$ and $W _{ br }=100 \ J$ respectively. The heat supplied to the system along the path iaf, ib and bf are $Q_{\mid a t l} Q_{b r}$ and $Q_{10}$ respectively. If the internal energy of the sytem in the state $b$ is $U_b=$ $200 \ J$ and $Q_{l a t}=500 \ J$, the ratio $Q_{b J} / Q_{10}$ is:View Solution
- 5One mole of an ideal monoatomic gas undergoes the following four reversible processes:View Solution
Step $1$ It is first compressed adiabatically from volume $V_{1}$ to $1 \;m ^{3}$.
Step $2$ Then expanded isothermally to volume $10 \;m ^{3}$.
Step $3$ Then expanded adiabatically to volume $V _{3}$.
Step $4$ Then compressed isothermally to volume $V_{1}$. If the efficiency of the above cycle is $3 / 4$, then $V_{1}$ is ............ $m^3$
- 6View SolutionWhich relation is correct for isometric process
- 7View SolutionThe adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
- 8An ideal heat engine working between temperature $T_1$ and $T_2 $ has an efficiency $\eta$, the new efficiency if both the source and sink temperature are doubled, will beView Solution
- 9An ideal gas is subjected to an isothermal expansion such that its volume changes from $V_i$ to $V_f$ and pressure from $P_i$ to $P_f$. The work done on the gas is :View Solution
- 10Two moles of an ideal monoatomic gas occupies a volume $V$ at $27^o C$. The gas expands adiabatically to a volume $2\ V$. Calculate $(a)$ the final temperature of the gas and $(b)$ change in its internal energy.View Solution
