The adiabatic Bulk modulus of a diatomic gas at atmospheric pressure is
Diffcult
We have the bulk modulus formula as$:$
Bulk Modulus $=\frac{\text { Pressure }}{\text { Strain }}=\frac{p}{\left(V_{0}-V_{n}\right) / V_{0}}$
For an adiabatic process, the bulk modulus is given by
$k=-\frac{V \Delta p}{\Delta V}=\gamma p$
adiabatic bulk modulus $=\gamma p$
At NTP, $p=1.013 \times 10^{5} N / m^{2}$ and $\gamma=1.4$
Hence Bulk modulus $=1.013 \times 10^{5} \times 1.4 \approx 1.4 \times 10^{5} N / m^{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas expands from volume $V_1$ to $V_2$. This may be achieved by either of the three processes: isobaric, isothermal and adiabatic. Let $\Delta U$ be the change in internal energy of the gas, $Q$ be the quantity of heat added to the system and $W$ be the work done by the system on the gas. Identify which of the following statements is false for $\Delta U$?View Solution
- 2The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
- 3Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
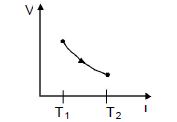
- 4The figure shows a process $AB$ undergone by $2$ moles of an ideal diatomic gas. The process $AB$ is in such a way that $VT =$ constant. $T_1 = 300 K $and $T_2 = 500 K$ ( $R = $ gas constant)View Solution
- 5View SolutionThe state of a thermodynamic system is represented by
- 6View SolutionOut of the following which quantity does not depend on path
- 7The pressure of an ideal gas varies with volume as $P = \alpha V,$ where $\alpha $ is a constant. One mole of the gas is allowed to undergo expansion such that its volume becomes $'m'$ times its initial volume. The work done by the gas in the process isView Solution
- 8A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
- 9One mole of a perfect gas in a cylinder fitted with a piston has a pressure $P,$ volume $V$ and temperature $T.$ If the temperature is increased by $1 \,K$ keeping pressure constant, the increase in volume isView Solution
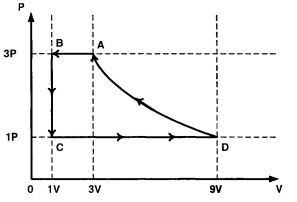
- 10One mole of a monatomic ideal gas is taken through a cycle $ABCDA$ as shown in the $P-V$ diagram. Column $II$ gives the characteristics involved in the cycle. Match them with each of the processes qiven in Column $I$View Solution
Column $I$ Column $II$ $(A)$ Process $A \rightarrow B$ $(p)$ Internal energy decreases. $(B)$ Process $B \rightarrow C$ $(q)$ Internal energy increases. $(C)$ Process $C \rightarrow D$ $(r)$ Heat is lost. $(D)$ Process $D \rightarrow A$ $(s)$ Heat is gained. $(t)$ Work is done on the gas.