$100\ g$ of water is heated from $30^o C$ to $50^o C$. Ignoring the slight expansion of the water, the change in its internal energy is .......$kJ$ (specific heat of water is $4184\ J/kg/K$):
AIEEE 2011, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
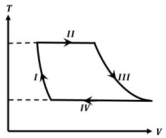
- 1One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
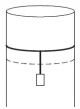
- 2A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
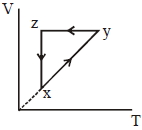
- 3A thermodynamic cycle $xyzx$ is shown on a $V-T$ diagram.View Solution
The $P-V$ diagram that best describes this cycle is
(Diagrams are schematic and not to scale)
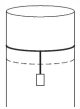
- 4A long cylindrical pipe of radius $20 \,cm$ is closed at its upper end and has an airtight piston of negligible mass as shown. When a $50 \,kg$ mass is attached to the other end of the piston, it moves down. If the air in the enclosure is cooled from temperature $T$ to $T-\Delta T$, the piston moves back to its original position. Then $\Delta T / T$ is close to (Assuming air to be an ideal gas, $g=10 \,m / s ^2$, atmospheric pressure is $10^5 \,Pa$ )View Solution
- 5View SolutionA cycle tyre bursts suddenly. This represents an
- 6View SolutionWhich of the following laws of thermodynamics defines the term internal energy?
- 7Two cylinders $A$ and $B$ of equal capacity are connected to each other via a stop cock. A contains an Ideal gas at standard temperature and pressure. $B$ is completely evacuated. The entire system is thermally insulated. The stop cock is suddenly opened. The process is :View Solution
- 8A Carnot engine has an efficiency of $50 \%$ when its source is at a temperature $327^{\circ}\,C$. The temperature of the sink is $.........^{\circ} C$View Solution
- 9A total of $48 \mathrm{~J}$ heat is given to one mole of helium kept in a cylinder. The temperature of helium increases by $2^{\circ} \mathrm{C}$. The work done by the gas is : (Given, $\mathrm{R}=8.3 \mathrm{~J} \mathrm{~K}^{-1} \mathrm{~mol}^{-1}$.)View Solution
- 10$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink areView Solution
