$A$ reversible engine converts one-sixth of the heat input into work. When the temperature of the sink is reduced by $ 62^oC$, the efficiency of the engine is doubled. The temperatures of the source and sink are
AIPMT 2000,AIEEE 2011,JEE MAIN 2019, Diffcult
(d) Initially $\eta = \left( {1 - \frac{{{T_2}}}{{{T_1}}}} \right) = \frac{W}{Q} = \frac{1}{6}$ ...$(i)$
Finally $\eta ' = \left( {1 - \frac{{{T_2}'}}{{{T_1}}}} \right) = \left( {1 - \frac{{({T_2} - 62)}}{{{T_1}}}} \right) = 1 - \frac{{{T_2}}}{{{T_1}}} + \frac{{62}}{{{T_1}}}$
$ = \eta + \frac{{62}}{{{T_1}}}$ ....$(ii)$
It is given that $\eta ' = 2\eta .$ Hence solving equation $ (i)$ and$(ii)$
==> ${T_1} = 372\,K = 99^\circ C$ and ${T_2} = 310K = 37^\circ C$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot engine, having an efficiency of $\eta = 1/10$ as heat engine, is used as a refrigerator. If the work done on the system is $10\ J$, the amount of energy absorbed from the reservoir at lower temperature is ....... $J$View Solution
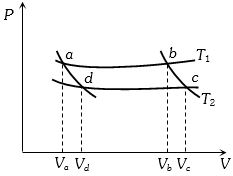
- 2In the following $P-V$ diagram two adiabatics cut two isothermals at temperatures $T_1$ and $T_2$ (fig.). The value of $\frac{{{V_a}}}{{{V_d}}}$ will beView Solution
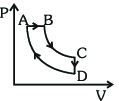
- 3A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution
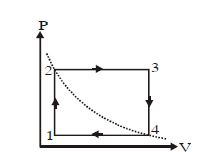
- 4One mole of an ideal gas undergoes a cyclic process, consisting of two isochores and two isobars. Temperature at $1$ and $3$ equal to $T_1$ and $T_3$ respectively. The work done by the gas over the cycle, if the point $2$ and $4$ lie on the same isothermView Solution
- 5Consider two containers $A$ and $B$ containing identical gases at the same pressure, volume and temperature. The gas in container $A$ is compressed to half of its original volume isothermally while the gas in container $B$ is compressed to half of its original value adiabatically. The ratio of final pressure of gas in $B$ to that of gas in $A$ isView Solution
- 6View SolutionA gas is compressed isothermally to half its initial volume. The same gas is compressed separately through an adiabatic process until its volume is again reduced to half. Then
- 7View SolutionIn Carnot engine the work done by working substance is equivalent to
- 8Given below are two statements : one is labelled as Assertion A and the other is labelled as Reason $R$. Assertion A : Efficiency of a reversible heat engine will be highest at $-273^{\circ} C$ temperature of cold reservoir.View Solution
Reason $R$ : The efficiency of Carnot's engine depends not only on temperature of cold reservoir but it depends on the temperature of hot reservoir too and is given as $\eta=\left(1-\frac{ T _2}{ T _1}\right)$.
In the light of the above statements, choose the correct answer from the options given below
- 9Volume versus temperature graph of two moles of helium gas is as shown in figure. The ratio of heat absorbed and the work done by the gas in process $1-2$ isView Solution
- 10A sample of gas expands from $V_1$ to $V _2$. In which of the following, the work done will be greatest ?View Solution