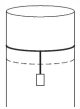
Initial pressure in cylinder is atmospheric pressure $p_0$.
When mass $m$ is attached to piston, then pressure $=p_0-\frac{m g}{A}$.
As, temperature remains constant during expansion
$\Rightarrow p_i V_i=p_f V_f$
$\Rightarrow p_0 V_i=\left(p_0-\frac{m g}{A}\right) \cdot V_f$
$\Rightarrow \frac{V_f}{V_i}=\frac{p_0}{\left(p_0-\frac{m g}{A}\right)}$
$\frac{V_i}{V_f}=1-\frac{m g}{p_0 A} \Rightarrow \frac{m g}{p_0 A}=1-\frac{V_i}{V_f}$
$\Rightarrow \frac{m g}{p_0 A}=V_f-V_i=\frac{\Delta V}{V_f}$
Now, when temperature is reduced by $\Delta T$, the volume of gas again contracts to its original volume.
$\Rightarrow \frac{V}{T}=$ constant
or $\quad \frac{\Delta V}{V}=\frac{\Delta T}{T}$
$\Rightarrow \frac{\Delta T}{T}=\frac{\Delta V}{V_f}=\frac{m g}{p_0 A}$
$\Rightarrow \frac{\Delta T}{T}=\frac{m g}{p_0 A}$
$=\frac{50 \times 10}{10^5 \times 3.14 \times(0.2)^2}$
$=\frac{5}{3.14 \times 4} \times \frac{10^2}{10^5 \times 10^{-2}}$
$=0.4 \times 10^{-1}=0.04$
Download our appand get started for free
Similar Questions
- 1$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 2An ideal gas expands in such a manner that its pressure and volume can be related by equation $P{V^2} = $ constant. During this process, the gas isView Solution
- 3'$n$' moles of an ideal gas undergoes a process $A \rightarrow B$ as shown in the figure. The maximum temperature of the gas during the process will beView Solution
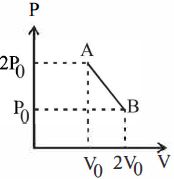
- 4View SolutionThe thermodynamic process, in which internal energy of the system remains constant is ...........
- 5Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram. Assuming the gas to be ideal the work done on the gas in taking it from $A $ to $B$ is ...... $R$View Solution

- 6A carnot engine with its cold body at $17\,^oC$ has $50\%$ effficiency. If the temperature of its hot body is now increased by $145\,^oC$, the efficiency becomes...... $\%$View Solution
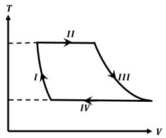
- 7One mole of a monatomic ideal gas undergoes a cyclic process as shown in the figure (where $V$ is the volume and $T$ is the temperature). Which of the statements below is (are) true?View Solution
(image)
$(A)$ Process $I$ is an isochoric process $(B)$ In process $II$, gas absorbs heat
$(C)$ In process $IV$, gas releases heat $(D)$ Processes $I$ and $III$ are $not$ isobaric
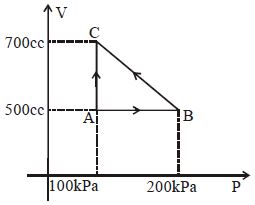
- 8A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 9A reversible engine has an efficiency of $\frac{1}{4}$. If the temperature of the sink is reduced by $58^{\circ} {C}$, its efficiency becomes double. Calculate the temperature of the sink. (In $^{\circ} {C}$)View Solution
- 10A thermally insulated rigid container contains an ideal gas heated by a filament of resistance $100 \,\Omega$ through a current of $1\,A$ for $5$ min then change in internal energy is...... $kJ$View Solution
