Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
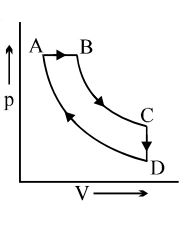
- 1A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processesView Solution
- 2The temperature of $3.00\, {mol}$ of an ideal diatomic gas is increased by $40.0^{\circ} {C}$ without changing the pressure of the gas. The molecules in the gas rotate but do not oscillate. If the ratio of change in internal energy of the gas to the amount of workdone by the gas is $\frac{{x}}{10} .$ Then the value of ${x}$ (round off to the nearest integer) is ..... . $\left(\right.$ Given $\left.{R}=8.31\, {J} {mol}^{-1} {K}^{-1}\right)$View Solution
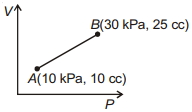
- 3Work done for the process shown in the figure is ............ $J$View Solution
- 4The pressure and density of a diatomic gas $(\gamma = 7/5)$ change adiabatically from $(P, d)$ to $(P', d')$. If $\frac{{d'}}{d} = 32$, then $\frac{{P'}}{P}$ should beView Solution
- 5A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
- 6$200\,g$ water is heated from $40\,^oC$ to $60\,^oC.$ Ignoring the slight expansion of water, the change in its internal energy is close to ...... $kJ$ (Given specific heat of water $=4184\,J/kgK$ )View Solution
- 7The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel isView Solution
- 8Determine coefficient of performance of given temperature limit.View Solution
$T_{1}=27^{\circ} C$ [outside fridge]
$T_{2}=-23^{\circ} C$ [inside fridge]
- 9Let $\eta_{1}$ is the efficiency of an engine at $T _{1}=447^{\circ}\,C$ and $T _{2}=147^{\circ}\,C$ while $\eta_{2}$ is the efficiency at $T _{1}=947^{\circ}\,C$ and $T _{2}=47^{\circ}\,C$. The ratio $\frac{\eta_{1}}{\eta_{2}}$ will be.View Solution
- 10One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
