$2\, kg$ of a monoatomic gas is at a pressure of $4\times10^4\, N/m^2$. The density of the gas is $8\, kg/m^3$. What is the order of energy of the gas due to its thermal motion ?
JEE MAIN 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Air is filled at $60^o C$ in a vessel of open mouth. The vessel is heated to a temperature $T$ so that $1/4^{th}$ part of air escapes. Assuming the volume of the vessel remaining constant, the value of $T$ is ....... $^oC$View Solution
- 2$50 \,cal$ of heat is required to raise the temperature of $1$ mole of an ideal gas from $20^{\circ} C$ to $25^{\circ} C$, while the pressure of the gas is kept constant. The amount of heat required to raise the temperature of the same gas through same temperature range at constant volume is ........ $cal$ $(R=2 \,cal / mol -K )$View Solution
- 3For a gas $\gamma = 7/5.$ The gas may probably beView Solution
- 4$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
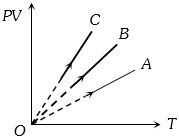
- 5According to the kinetic theory of gases the $r.m.s.$ velocity of gas molecules is directly proportional toView Solution
- 6Number of molecules in a volume of $4\, cm ^{3}$ of a perfect monoatomic gas at some temperature $T$ and at a pressure of $2\, cm$ of mercury is close to $?$View Solution
(Given, mean kinetic energy of a molecule (at $T$ ) is $4 \times 10^{-14}$ erg, $g=980\, cm / s ^{2}$, density of mercury $=13.6\, g / cm ^{3}$)
- 7View SolutionOne mole of a monoatomic ideal gas is mixed with one mole of a diatomic ideal gas. The molar specific heat of the mixture at constant volume is
- 8The given diagram shows isotherms for a fixed mass of an ideal gas at temperature $T_1$ and $T_2$ . What is the value of the ratio $\frac{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_{2{\text{ }}}}}}{{{\text{r}}{\text{.m}}{\text{.s}}{\text{. speed of the molecules at temperature }}{{\text{T}}_1}}}$ ?View Solution
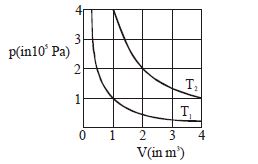
- 9Vessel $A$ is filled with hydrogen while vessel $B,$ whose volume is twice that of $A,$ is filled with the same mass of oxygen at the same temperature. The ratio of the mean kinetic energies of hydrogen and oxygen isView Solution
- 10The value of $\gamma\left(=\frac{\mathrm{C}_{\mathrm{p}}}{\mathrm{C}_{\mathrm{v}}}\right),$ for hydrogen, hellium and another ideal diatomic gas $X$(whose molecules are not rigid but have an additional vibrational mode), are respectively equal toView Solution
