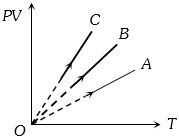
$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternative

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA real gas behaves like an ideal gas if its
- 2An ideal gas filled in a cylinder occupies volume $V$. The gas is compressed isothermally to the volume $V/3$. Now, the cylinder valve is opened and the gas is allowed to leak keeping temperature same. What percentage of the number of molecules should escape to bring the pressure in the cylinder back to its original value?View Solution
- 3The volume of a gas will be double of what it is at $0°C$ (pressure remaining constant) atView Solution
- 4One mole of monoatomic gas and three moles of diatomic gas are put together in a container. The molar specific heat (in $J\,{K^{ - 1}}\,mo{l^{ - 1}})$ at constant volume is $(R = 8.3\,J\,{K^{ - 1}}\,mo{l^{ - 1}})$View Solution
- 5The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\, cc$ at sea level (pressure $1$ atmosphere, temperature $27°C$). Assuming that the temperature around Mount Everest is $-73°C$ and that the oxygen cylinder has capacity of $5.2 \,litre,$ the pressure at which ${O_2}$ be filled (at site) in cylinder is .... $atm$View Solution
- 6Two closed containers of equal volume filled with air at pressure $P_0$ and temperature $T_0$. Both are connected by a narrow tube. If one of the container is maintained at temperature $T_0$ and other at temperature $T$, then new pressure in the containers will beView Solution
- 7For a certain gas, the ratio of specific heats is given to be $\gamma = 1.5$. For this gasView Solution
- 8The speeds of $5$ molecules of a gas (in arbitrary units) are as follows : $2, 3, 4, 5, 6.$ The root mean square speed for these molecules isView Solution
- 9The relation between the gas pressure $P$ and average kinetic energy per unit volume $E$ isView Solution
- 10$2\, kg$ of a monoatomic gas is at a pressure of $4\times10^4\, N/m^2$. The density of the gas is $8\, kg/m^3$. What is the order of energy of the gas due to its thermal motion ?View Solution
