The value of $\gamma\left(=\frac{\mathrm{C}_{\mathrm{p}}}{\mathrm{C}_{\mathrm{v}}}\right),$ for hydrogen, hellium and another ideal diatomic gas $X$(whose molecules are not rigid but have an additional vibrational mode), are respectively equal to
NEET 2019, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Given below are two statements :View Solution
Statement $(I)$ : The mean free path of gas molecules is inversely proportional to square of molecular diameter.
Statement $(II)$ : Average kinetic energy of gas molecules is directly proportional to absolute temperature of gas.
In the light of the above statements, choose the correct answer from the option given below:
- 2View SolutionThe root mean square speed of the molecules of a gas is
- 3The average distance between molecules of an ideal gas at $STP$ is approximately of the order ofView Solution
- 4A cylinder made of perfectly non conducting material closed at both ends is divided into two equal parts by a heat proof piston. Both parts of the cylinder contain the same masses of a gas at a temperature $t_0 = 27^o$ and pressure $P_0 = 1$ atm. Now if the gas in one of the parts is slowly heated to $t = 57^oC$ while the temperature of first part is maintained at $t_0$ the distance moved by the piston from the middle of the cylinder will be.... $cm$ (length of the cylinder $= 84\,\, cm$)View Solution
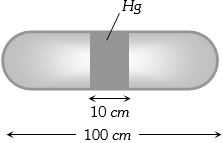
- 5A horizontal uniform glass tube of $100 \,cm$, length sealed at both ends contain $10 \,cm$ mercury column in the middle. The temperature and pressure of air on either side of mercury column are respectively $81°C$ and $76\, cm$ of mercury. If the air column at one end is kept at $0°C$ and the other end at $273°C$, the pressure of air which is at $0°C$ is (in $cm$ of $Hg$)View Solution
- 6A flask contains argon and oxygen in the ratio of $3: 2$ in mass and the mixture is kept at $27^{\circ} C$. The ratio of their average kinetic energy per molecule respectively will be ...........View Solution
- 7Find $\gamma$ for the mixture of $11\, gm CO _{2}$ and $14\, gm N _{2} ?$View Solution
- 8A mixture of ideal gases has $2$ moles of He, $4$ moles of oxygen and $1$ mole of ozone at absolute temperature $T$. The internal energy of mixture is ............. $RT$View Solution
- 9At temperature $T,$ the $r.m.s.$ speed of helium molecules is the same as $r.m.s.$ speed of hydrogen molecules at normal temperature and pressure. The value of $T$ is ....... $^oC$View Solution
- 10Let $\gamma_1$ be the ratio of molar specific heat at constant pressure and molar specific heat at constant volume of a monoatomic gas and $\gamma_2$ be the similar ratio of diatomic gas. Considering the diatomic gas molecule as a rigid rotator, the ratio, $\frac{\gamma_1}{\gamma_2}$ isView Solution
