A balloon carries a total load of $185\; {kg}$ at normal pressure and temperature of $27^{\circ} {C}$. What load will the balloon carry on rising to a height at which the barometric pressure is $45\; {cm}$ of ${Hg}$ and the temperature is $-7^{\circ} {C}$. Assuming the volume constant? (in ${kg}$)
JEE MAIN 2021, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas in a closed container is slowly heated. As its temperature increases, which of the following statements are true$?$View Solution
$(A)$ the mean free path of the molecules decreases.
$(B)$ the mean collision time between the molecules decreases.
$(C)$ the mean free path remains unchanged.
$(D)$ the mean collision time remains unchanged.
- 2View SolutionMean kinetic energy (or average energy) per gm molecule of a monoatomic gas is given by
- 3A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
- 4View SolutionAn ant is moving on a plane horizontal surface. The number of degrees of freedom of the ant will be .........
- 5To what temperature should the hydrogen at $327°C$ be cooled at constant pressure, so that the root mean square velocity of its molecules become half of its previous value ....... $^oC$View Solution
- 6The average kinetic energy of hydrogen molecules at $300 K$ is $E.$ At the same temperature, the average kinetic energy of oxygen molecules will beView Solution
- 7Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
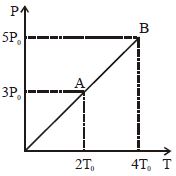
- 8In the isothermal expansion of $10\,g$ of gas from volume $V$ to $2V$ the work done by the gas is $575\,J$. What is the root mean square speed of the molecules of the gas at that temperature ..... $m/s$?View Solution
- 9View SolutionThe degrees of freedom of a stationary rigid body about its axis will be
- 10View SolutionAccording to kinetic theory of gases,
