The degrees of freedom of a stationary rigid body about its axis will be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Which of the following shows the correct relationship between the pressure $'P'$ and density $\rho $ of an ideal gas at constant temperature ?View Solution
- 2A diatomic gas molecule has translational, rotational and vibrational degrees of freedom. The ${C_P}/{C_V}$ isView Solution
- 3What is the value of $\frac{R}{{{C_P}}}$ for diatomic gasView Solution
- 4A certain amount of an ideal monatomic gas needs $20 \,J$ of heat energy to raise its temperature by $10^{\circ} C$ at constant pressure. The heat needed for the same temperature rise at constant volume will be ........ $J$View Solution
- 5For the $P-V$ diagram given for an ideal gas, out of the following which one correctly represents the $T-P$ diagram ?View Solution
- 6$7$ mole of certain monoatomic ideal gas undergoes a temperature increase of $40 K$ at constant pressure. The increase in the internal energy of the gas in this process is$....J$ (Given $R =8.3 JK ^{-1} mol ^{-1}$ )View Solution
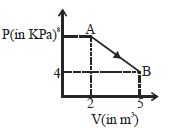
- 7A mole of an ideal diatomic gas undergoes a transition from $A$ to $B$ along a path $AB$ as shown in the figure, The change in internal energy of the gas during the transition is ...... $kJ$View Solution
- 8At what temperature volume of an ideal gas at $0°C$ becomes triple ....... $^oC$View Solution
- 9View SolutionIndicator diagram is a ...........
- 10View SolutionThe specific heat of a gas
