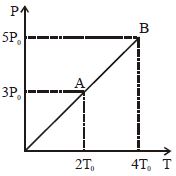
Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will be

Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas undergoes a quasi static, reversible process in which its molar heat capacity $C$ remains constant. If during this process the relation of pressure $P$ and volume $V$ is given by $PV^n =$ constant, then n is given by (Here $C_p$ and $C_v$ are molar specific heat at constant pressure and constant volume, respectively) :View Solution
- 2In a thermally isolated system, two boxes filled with an ideal gas are connected by a valve. When the valve is in closed position, states of the box $1$ and $2$ respectively, are ( $1 \,atm , V, T)$ and $(0.5 \,atm , 4 V, T)$. When the valve is opened, then the final pressure of the system is approximately ............... $atm$View Solution
- 3The temperature of an ideal gas is increased from $200\,K$ to $800\,K$. If r.m.s. speed of gas at $200\,K$ is $v_0$. Then, r.m.s. speed of the gas at $800\,K$ will be:View Solution
- 4If one mole of the polyatomic gas is having two vibrational modes and $\beta$ is the ratio of molar specific heats for polyatomic gas $\left(\beta=\frac{ C _{ P }}{ C _{ v }}\right)$ then the value of $\beta$ is:View Solution
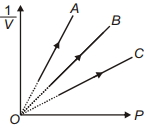
- 5Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
- 6An air bubble of volume $1.0\; cm ^{3}$ rises from the bottom of a lake $40\; m$ deep at a temperature of $12\,^{\circ} C$. To what volume (in $cm^3$) does it grow when it reaches the surface, which is at a temperature of $35\,^{\circ} C ?$View Solution
- 7The temperature at which the root mean square velocity of a molecule will be doubled than at $100°C$View Solution
- 8A monoatomic gas performs a work of $\frac{Q}{4}$ where $Q$ is the heat supplied to it. The molar heat capaticy of the gas will be $R$ during this transformation. Where $R$ is the gas constant.View Solution
- 9View SolutionThe vapour of a substance behaves as a gas
- 10An ideal gas has molecules with $5\,$ degrees of freedom. The ratio of specific heats at constant pressure $(C_p)$ and at constant volume $(C_v)$ isView Solution
