According to kinetic theory of gases,
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The average degree of freedom per molecule of a gas is $6.$ The gas performs $25 \,J$ work, while expanding at constant pressure. The heat absorbed by the gas is ...... $J$View Solution
- 2An $HCl$ molecule has rotational, translational and vibrational motions. If the $rms$ velocity of $HCl$ molecules in its gaseous phase is $\bar v ,\,m$ is its mass and $k_B$ is Boltzmann constant, then its temperature will beView Solution
- 3View SolutionThe molecules of an ideal gas at a certain temperature have
- 4The number of air molecules per $\mathrm{cm}^3$ increased from $3 \times 10^{19}$ to $12 \times 10^{19}$. The ratio of collision frequency of air molecules before and after the increase in number respectively is $.........$View Solution
- 5View SolutionThe vapour of a substance behaves as a gas
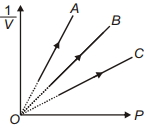
- 6Figure shows the isotherms of a fixed mass of an ideal gas at three temperatures $T_A, T_B$ and $T_C$, thenView Solution
- 7The specific heats, $C_P$ and $C_V$ of a gas of diatomic molecules, $A$, are given (in units of $J\, mol^{-1}\, K^{-1}$) by $29$ and $22$, respectively. Another gas of diatomic molecules $B$, has the corresponding values $30$ and $21$. If they are treated as ideal gases, thenView Solution
- 8Let $\bar v,\;{v_{rms}}$ and ${v_p}$ respectively denote the mean speed, root mean square speed and most probable speed of the molecules in an ideal monoatomic gas at absolute temperature $T.$ The mass of a molecule is $m.$ ThenView Solution
- 9At a temperature $T K,$ the pressure of $4.0\,\,g$ argon in a bulb is $p$. The bulb is put in a bath having temperature higher by $50\,\,K$ than the first one. $0.8\,\,g$ of argon gas had to be removed to maintained original pressure. The temperature $T$ is equal to ..... $K$View Solution
- 10A resistance coil connected to an external battery is placed inside an adiabatic cylinder fitted with a frictionless pistn and containing an ideal gas. A current $i$ flows through the coil which has a resistance $R$. At what speed must the piston move upward in order that the temperature of the gas remains uchanged? Neglect atmospheric pressure.View Solution