A Carnot engine operating between two reservoirs has efficiency $\frac{1}{3}$. When the temperature of cold reservoir raised by $x$, its efficiency decreases to $\frac{1}{6}$. The value of $x$, if the temperature of hot reservoir is $99^{\circ}\,C$, will be $........\,K$
JEE MAIN 2023, Medium
$T _{ H }=99^{\circ} C =99+273$
$\qquad=372\,K$
$1-\frac{ T _{ C }}{ T _{ H }}=\frac{1}{3}$
$\frac{ T _{ C }}{ T _{ H }}=\frac{2}{3} \quad(1) \Rightarrow T _{ C }=\frac{2}{3} \times 372$
$1-\frac{ T _{ C }+ X }{ T _{ H }}=\frac{1}{6}$
$\frac{5}{6}=\frac{ T _{ C }+ X }{ T _{ H }}$
$\frac{5}{6}=\frac{248+ X }{372}$
$248+ X =5 \times 62$
$X =310-248=62\,K$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A rigid diatomic ideal gas undergoes an adiabatic process at room temperature. The rational between temperature and volume for the process is $TV^x =$ constant, then $x$ isView Solution
- 2The $P-V$ diagram of a system undergoing thermodynamic transformation is shown in figure. The work done by the system in going from $A \to B \to C$ is $30J$ and $40J$ heat is given to the system. The change in internal energy between $A$ and $C$ is ....... $J$View Solution
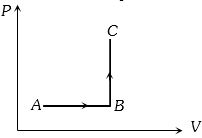
- 3View SolutionDuring the thermodynamic process shown in figure for an ideal gas
- 4A Carnot engine operates between ${227^o}C$ and ${27^o}C.$ Efficiency of the engine will beView Solution
- 5Two moles of an ideal monoatomic gas occupies a volume $V$ at $27^o C$. The gas expands adiabatically to a volume $2\ V$. Calculate $(a)$ the final temperature of the gas and $(b)$ change in its internal energy.View Solution
- 6View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
- 7Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)View Solution
- 8The pressure $P_{1}$ and density $d_{1}$ of diatomic gas $\left(\gamma=\frac{7}{5}\right)$ changes suddenly to $P _{2}\left(> P _{1}\right)$ and $d _{2}$ respectively during an adiabatic process. The temperature of the gas increases and becomes $......$ times of its initial temperature.$\left(\right.$ given $\left.\frac{ d _{2}}{ d _{1}}=32\right)$View Solution
- 9Considere the thermodynamics cycle shown on $PV$ diagram. The process $A \rightarrow B$ is isobaric, $B \rightarrow C$ is isochoric and $C \rightarrow A$ is a straight line process. The following internal energy and heat are given $: \Delta U_{A \rightarrow B} = + 400\,\, kJ$ and $Q_{B \rightarrow C} = - 500\,\, kJ$ The heat flow in the process $Q_{C \rightarrow A}$ is ...... $kJ$View Solution
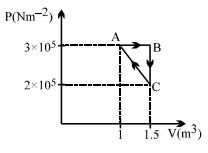
- 10In thermodynamic process, $200$ Joules of heat is given to a gas and $100$ Joules of work is also done on it. The change in internal energy of the gas is ........ $J$View Solution
