Half mole of an ideal monoatomic gas is heated at constant pressure of $1\, atm$ from $20\,^oC$ to $90\,^oC$. Work done by has is close to ..... $J$ (Gas constant $R = 8.31\, J/mol.K$)
JEE MAIN 2019, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1The volume $V$ of a given mass of monoatomic gas changes with temperature $T$ according to the relation $V = KT ^{2 / 3}$. The workdone when temperature changes by $90\, K$ will be $x\,R$. The value of $x$ is $[ R =$ universal gas constant $]$View Solution
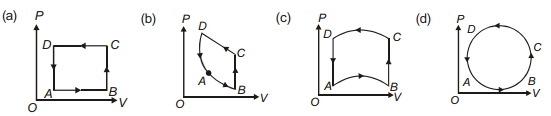
- 2In following figures $(a)$ to $(d)$, variation of volume by change of pressure is shown in figure. The gas is taken along the path $A B C D A$. Change in internal energy of the gas will be .......View Solution
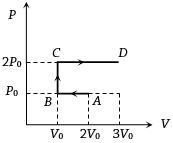
- 3$P-V$ diagram of an ideal gas is as shown in figure. Work done by the gas in process $ABCD$ isView Solution
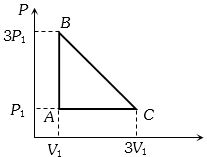
- 4An ideal gas is taken around the cycle $ABCA$ as shown in the $P-V $ diagram. The net work done by the gas during the cycle is equal toView Solution
- 5An ideal monoatomic gas with pressure $P$, volume $V$ and temperature $T$ is expanded isothermally to a volume $2\, V$ and a final pressure $P_i$. If the same gas is expanded adiabatically to a volume $2\,V$, the final pressure is $P_a$ . The ratio $\frac{{{P_a}}}{{{P_i}}}$ isView Solution
- 6An ideal gas heat engine operates in Carnot cycle between $227°C$ and $127°C.$ It absorbs $6 \times {10^4}$ cals of heat at higher temperature. Amount of heat converted to work is .........$ \times {10^4}\; cal$View Solution
- 7One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
- 8View SolutionWhen heat in given to a gas in an isobaric process, then
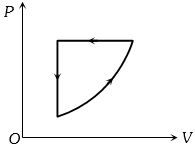
- 9For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution
- 10When the amount of work done is $333\, cal$ and change in internal energy is $167\, cal$, then the heat supplied is ....... $cal$View Solution
