A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$
Medium
(d) $\eta = \frac{{{T_1} - {T_2}}}{{{T_1}}} = \frac{W}{Q}$
==> $Q = \left( {\frac{{{T_1}}}{{{T_1} - {T_2}}}} \right)\,W$
$ = \frac{{600}}{{(600 - 300)}} \times 800 =1600 \,J$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionIn which of the following processes, heat is neither absorbed nor released by a system ?
- 2A thermally insulated container is divided into two parts by a screen. In one part the pressure and temperature are $P$ and $T$ for an ideal gas filled. In the second part it is vacuum. If now a small hole is created in the screen, then the temperature of the gas willView Solution
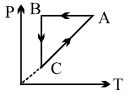
- 3A cyclic process $ABCA$ is shown in $PT$ diagram. When presented on $PV$, it wouldView Solution
- 4A gas at initial temperature $T$ undergoes sudden expansion from volume $V$ to $2 \,V$. Then,View Solution
- 5One mole of an ideal gas expands adiabatically from an initial state $\left(T_A, V_0\right)$ to final state $\left(T_f, 5 V_0\right)$. Another mole of the same gas expands isothermally from a different initial state ( $T_{\mathrm{B}}, \mathrm{V}_0$ ) to the same final state $\left(T_{\mathrm{f}}, 5 V_0\right)$. The ratio of the specific heats at constant pressure and constant volume of this ideal gas is $\gamma$. What is the ratio $T_{\mathrm{A}} / T_{\mathrm{B}}$ ?View Solution
- 6Determine efficiency of carnot cycle if in adiabatic expansion volume $3$ times of initial value and $\gamma =1.5$View Solution
- 7Adiabatic modulus of elasticity of a gas is $2.1 \times {10^5}N/{m^2}.$ What will be its isothermal modulus of elasticity $\left( {\frac{{{C_p}}}{{{C_v}}} = 1.4} \right)$View Solution
- 8The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 9In an adiabatic process where in pressure is increased by $\frac{2}{3}\% $ if $\frac{{{C_p}}}{{{C_v}}} = \frac{3}{2},$ then the volume decreases by aboutView Solution
- 10A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
