A Carnot's engine works as a refrigerator between $250\, K$ and $300\, K$. It receives $500\, cal$ heat from the reservoir at the lower temperature. The amount of work done in each cycle to operate the refrigerator is ..... $J$
JEE MAIN 2018, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
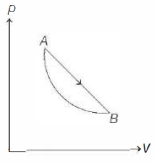
- 1An ideal gas is made to undergo a cycle depicted by the $p-V$ diagram given below. The curved line from $A$ to $B$ is an adiabat.Then,View Solution
- 2A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
- 3Find the change in the entropy in the following process $100 \,gm$ of ice at $0°C$ melts when dropped in a bucket of water at $50°C$ (Assume temperature of water does not change) ..... $ cal/K$View Solution
- 4$n-$ $moles$ of an ideal gas with constant volume heat capacity $C_v$ undergo an isobaric expansion by certain volume. The ratio of the work done in the process, to the heat supplied isView Solution
- 5View SolutionIn a cyclic process, the internal energy of the gas
- 6Work done in the given $P-V$ diagram in the cyclic process isView Solution
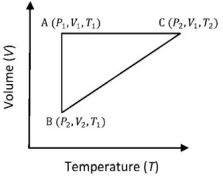
- 7A reversible cyclic process for an ideal gas is shown below. Here, $P, V$, and $T$ are pressure, volume and temperature, respectively. The thermodynamic parameters $q, w, H$ and $U$ are heat, work, enthalpy and internal energy, respectively.View Solution
(image)
The correct option ($s$) is (are)
$(A)$ $q_{A C}=\Delta U_{B C}$ and $W_{A B}=P_2\left(V_2-V_1\right)$
$(B)$ $\mathrm{W}_{\mathrm{BC}}=\mathrm{P}_2\left(\mathrm{~V}_2-\mathrm{V}_1\right)$ and $\mathrm{q}_{\mathrm{BC}}=\mathrm{H}_{\mathrm{AC}}$
$(C)$ $\Delta \mathrm{H}_{\mathrm{CA}}<\Delta \mathrm{U}_{\mathrm{CA}}$ and $\mathrm{q}_{\mathrm{AC}}=\Delta \mathrm{U}_{\mathrm{BC}}$
$(D)$ $\mathrm{q}_{\mathrm{BC}}=\Delta \mathrm{H}_{\mathrm{AC}}$ and $\Delta \mathrm{H}_{\mathrm{CA}}>\Delta \mathrm{U}_{\mathrm{CA}}$
- 8View SolutionOne mole of a monoatomic gas and one mole of a diatomic gas are initially in the same state. Both gases are expanded isothermally and then adiabatically, such that they acquire the same final state. Choose the correct statement.
- 9In changing the state of thermodynamics from $A$ to $B$ state, the heat required is $Q$ and the work done by the system is $W.$ The change in its internal energy isView Solution
- 10For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correctView Solution