For one complete cycle of a thermodynamic process on a gas as shown in the $P-V$ diagram, Which of following is correct
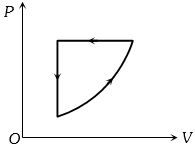

Easy
(a) $\Delta {E_{{\rm{int}}}} = 0$,
for a complete cycle and for given cycle work done is negative,
so from first law of thermodynamics $Q$ will be negative i.e. $Q < 0$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A thermodynamic system goes from states $(i)\,\,{P_1}, V$ to $2{P_1}, V\, (ii)\, P, V$ to $P, 2V.$ Then work done in the two cases isView Solution
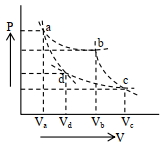
- 2Two different adiabatic paths for the same gas intersect two isothermal curves as shown in$P-V$ diagram. The relation between the ratio $\frac{V_a}{V_d}$ and the ratio $\frac{V_b}{V_c}$ is:View Solution
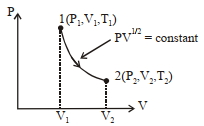
- 3Thermodynamic process is shown below on a $P-V$ diagram for one mole of an ideal gas. If $V _{2}=2 V _{1}$ then the ratio of temperature $T _{2} / T _{1}$ is ...... .View Solution
- 4$Assertion :$ Thermodynamic process in nature are irreversible.View Solution
$Reason :$ Dissipative effects cannot be eliminated. - 5View SolutionWhen heat in given to a gas in an isobaric process, then
- 6This question has Statement $1$ and Statement $2.$ Of the four choices given after the Statements, choose the one that best describes the two Statements.View Solution
Statement $1 :$ An inventor claims to have constructed an engine that has an efficiency of $30\%$ when operated between the boiling and freezing points of water. This is not possible.
Statement $2:$ The efficiency of a real engine is always less than the efficiency of a Carnot engine operating between the same two temperatures.
- 7The temperature of reservoir of Carnot's engine operating with an efficiency of $70\%$ is $1000K.$ The temperature of its sink is ...... $K$View Solution
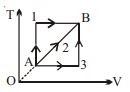
- 8A given mass of a gas expands from a state $A$ to the state $B$ by three paths $1, 2$ and $3$ as shown in $T-V$ indicator diagram. If $W_1, W_2$ and $W_3$ respectively be the work done by the gas along the three paths, thenView Solution
- 9A thermodynamic cycle takes in heat energy at a high temperature and rejects energy at a lower temperature. If the amount of energy rejected at the low temperature is $3$ times the amount of work done by the cycle, the efficiency of the cycle isView Solution
- 10View SolutionThe process in which no heat enters or leaves the system is termed as
