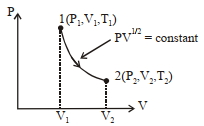
A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
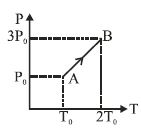
- 1Pressure versus temperature graph of an ideal gas is as shown in figure. Density of the gas at point $A$ is ${\rho _0}$. Density at point $B$ will beView Solution
- 2Match List$-I$ with List$-II$View Solution
List$-I$ List$-II$ $(a)$ Isothermal $(i)$ Pressure constant $(b)$ Isochoric $(ii)$ Temperature constant $(c)$ Adiabatic $(iii)$ Volume constant $(d)$ Isobaric $(iv)$ Heat content is constant Choose the correct answer from the options given below
- 3A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
- 4View SolutionWhen a gas expands adiabatically
- 5One mole of an ideal gas at initial temperature $T$, undergoes a quasi-static process during which the volume $V$ is doubled. During the process, the internal energy $U$ obeys the equation $U=a V^3$, where $a$ is a constant. The work done during this process isView Solution
- 6A diatomic gas $(\gamma=1.4)$ does $200 \mathrm{~J}$ of work when it is expanded isobarically. The heat given to the gas in the process is:View Solution
- 7$540$ calories of heat convert $1 $ cubic centimeter of water at ${100^o}C$ into $1671 $ cubic centimeter of steam at ${100^o}C$ at a pressure of one atmosphere. Then the work done against the atmospheric pressure is nearly ...... $cal$View Solution
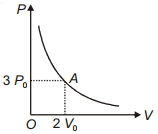
- 8The variation of pressure $P$ with volume $V$ for an ideal monatomic gas during an adiabatic process is shown in figure. At point $A$ the magnitude of rate of change of pressure with volume isView Solution
- 9$Assertion :$ In adiabatic compression, the internal energy and temperature of the system get decreased.View Solution
$Reason :$ The adiabatic compression is a slow process. - 10Thermodynamic process is shown below on a $P-V$ diagram for one mole of an ideal gas. If $V _{2}=2 V _{1}$ then the ratio of temperature $T _{2} / T _{1}$ is ...... .View Solution