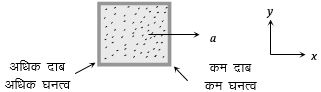
A closed compartment containing gas is moving with some acceleration in horizontal direction. Neglect effect of gravity. Then the pressure in the compartment is
IIT 1999, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionFor ideal gas, which statement is not true
- 2A cylinder of fixed capacity $44.8$ litre. contains a monatomic gas at standard temperature and pressure. The amount of heat required to cylinder by $10^o C$ will be. ($R=$ universal gas constant)View Solution
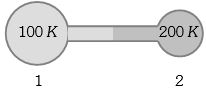
- 3Figure shows two flasks connected to each other. The volume of the flask $1$ is twice that of flask $2.$ The system is filled with an ideal gas at temperature $100\, K$ and $200 \,K $ respectively. If the mass of the gas in $1$ be $m$ then what is the mass of the gas in flask $2$View Solution
- 4$N( < 100)$ molecules of a gas have velocities $1, 2, 3\,.\,.\,.\,.\,.N/km/s$ respectively. ThenView Solution
- 5The pressure of an ideal gas is written as $E = \frac{3PV}{2}$ . Here $E$ stands forView Solution
- 6If $10^{22}$ gas molecules each of mass $10^{-26}\, kg$ collide with a surface (perpendicular to it)elastically per second over an area $1\, m^2$ with a speed $10^4\,m/s$, the pressure exerted by the gas molecules will be of the order ofView Solution
- 7View SolutionThe pressure exerted by the gas on the walls of the container because
- 8The air density at Mount Everest is less than that at the sea level. It is found by mountaineers that for one trip lasting a few hours, the extra oxygen needed by them corresponds to $30,000\, cc$ at sea level (pressure $1$ atmosphere, temperature $27°C$). Assuming that the temperature around Mount Everest is $-73°C$ and that the oxygen cylinder has capacity of $5.2 \,litre,$ the pressure at which ${O_2}$ be filled (at site) in cylinder is .... $atm$View Solution
- 9An $HCl$ molecule has rotational, translational and vibrational motions. If the $rms$ velocity of $HCl$ molecules in its gaseous phase is $\bar v ,\,m$ is its mass and $k_B$ is Boltzmann constant, then its temperature will beView Solution
- 10The temperature of $5$ moles of a gas which was held at constant volume was changed from ${100^o}C$ to ${120^o}C$. The change in internal energy was found to be $80$ Joules. The total heat capacity of the gas at constant volume will be equal to ...... $J/K$View Solution