A cube of ice has a iron piece fronzen unside at. The cube floats un a beakerfilled with water, when the ice melts then, the level of water in beaker.
Easy
Consider an ice cube which floats on a beaker filled with water, containing an ison piece as shown.
Let $V$ be the volume $g$ water. displaced by ice due to mass.
$V=$ volume og water displaced.
$m_1=$ mass of ice
$m_2=$ mass of iron pitce,
$\rho_1=$ density of water,
$\rho_2=$ density of iron piece.
Now, $v=\frac{\left(m_1+m_2\right)}{\rho_1} \quad[$ before melting $]$.
And, $\quad V^{\prime}=\frac{m_1}{\rho_1}+\frac{m_2}{\rho_2} \quad[$ after melting $]$.
Since $\rho_2 > \rho_1$. So, $\quad \frac{m_2}{\rho_2} < \frac{m_2}{\rho_1}$.
Thus, $\quad v > V^{\prime}$
So, when the whole ice melts, the volume of water decreases.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A capillary tube is attached horizontally to a constant head arrangement. If the radius of the capillary tube is increased by $10\%$ then the rate of flow of liquid will change nearly by ......... $\%$View Solution
- 2View SolutionVelocity of water in a river is
- 3Water is flowing in a pipe of diameter $ 4 cm$ with a velocity $3 m/s. $ The water then enters into a tube of diameter $ 2 cm.$ The velocity of water in the other pipe is .......... $m/s$View Solution
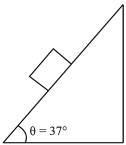
- 4A cubical block of side $‘a’$ and density $‘\rho ’$ slides over a fixed inclined plane with constant velocity $‘v’$. There is a thin film of viscous fluid of thickness $‘t’$ between the plane and the block. Then the coefficient of viscosity of the thin film will be:View Solution
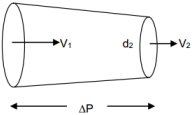
- 5Determine the pressure difference in tube of non$-$uniform cross sectional area as shown in figure. $\Delta P =?$ (in $pa$)View Solution
$d_{1}=5\, cm , V_{1}=4\, cm , d_{2}=2\, cm , V_{2}=?$
- 6A liquid of density $\rho $ is coming out of a hose pipe of radius $a$ with horizontal speed $v$ and hits a mesh. $50\%$ of the liquid passes through the mesh unaffected. $25\%$ looses all of its momentum and $25\%$ comes back with the same speed. The resultant pressure on the mesh will beView Solution
- 7A small spherical ball of radius $r$, falling through a viscous medium of negligible density has terminal velocity ' $v$ '. Another ball of the same mass but of radius $2 r$, falling through the same viscous medium will have terminal velocity:View Solution
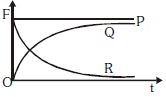
- 8A heavy spherical ball is dropped near surface in a long column of viscous liquid. Which of the following graphs represent the variation ofView Solution
$(i)$ Gravitational force with time
$(ii)$ Viscous force with time
$(iii)$ Net force acting on the ball with time
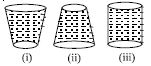
- 9View SolutionThe three water filled tanks shown have the same volume and height. If small identical holes are punched near this bottom, which one will be the first to get empty.
- 10A hot air balloon is carrying some passengers, and a few sandbags of mass $1 kg$ each so that its total mass is $480 kg$. Its effective volume giving the balloon its buoyancy is $V$. The balloon is floating at an equilibrium height of $100 m$. When $N$ number of sandbags are thrown out, the balloon rises to a new equilibrium height close to $150 m$ with its volume $V$ remaining unchanged. If the variation of the density of air with height $h$ from the ground is $\rho(h)=\rho_0 e^{-\frac{h}{h_0}}$, where $\rho_0=1.25 kg m ^{-3}$ and $h _0=6000 m$, the value of $N$ is. . . . .View Solution
