A small spherical ball of radius $r$, falling through a viscous medium of negligible density has terminal velocity ' $v$ '. Another ball of the same mass but of radius $2 r$, falling through the same viscous medium will have terminal velocity:
JEE MAIN 2024, Diffcult
Since density is negligible hence Buoyancy force will be negligibleAt terminal velocity.
${Mg}=6 \pi \eta \mathrm{rv}$
$\mathrm{V} \propto \frac{1}{\mathrm{r}} \quad$ (as mass is constant)
Now, $\frac{\mathrm{v}}{\mathrm{v}^{\prime}}=\frac{\mathrm{r}^{\prime}}{\mathrm{r}}$
$r^{\prime}=2 \mathrm{r}$
So, $v^{\prime}=\frac{v}{2}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two drops of same radius are falling through air with steady velocity of $v $ $cm/s$. If the two drops coalesce, what would be the terminal velocity?View Solution
- 2View SolutionThe property of a liquid by which it opposes the flow of itself is called ..........
- 3Mercury is filled in a tube of radius $2 \mathrm{~cm}$ up to a height of $30 \mathrm{~cm}$. The force exerted by mercury on the bottom of the tube is. . . . . . $\mathrm{N}$.View Solution
(Given, atmospheric pressure $=10^5 \mathrm{Nm}^{-2}$, density of mercury $=1.36 \times 10^4 \mathrm{~kg} \mathrm{~m}^3, \mathrm{~g}=10 \mathrm{~ms}^2$, $\left.\pi=\frac{22}{7}\right)$
- 4Figure below shows a liquid being pushed out of the tube by a piston having area of cross section $2.0\,cm ^2$. The area of cross section at the outlet is $10\,mm ^2$. If the piston is pushed at a speed of $4\,cm s ^{-1}$, the speed of outgoing fluidis $.........\,cm s ^{-1}$.View Solution

- 5An ornament weighs $10\, g$ in air and $6\, g$ in water. Density of material of ornament is $20\,\frac{g}{{cc}}$. The volume of cavity in ornament is ........ $cc$View Solution
- 6A $U-$ tube in which the cross-sectional area of the limb on the left is one quarter, the limb on the right contains mercury (density $13.6 g/cm^3$). The level of mercury in the narrow limb is at a distance of $36 cm$ from the upper end of the tube. What will be the rise in the level of mercury in the right limb if the left limb is filled to the top with water ........ $cm$View Solution
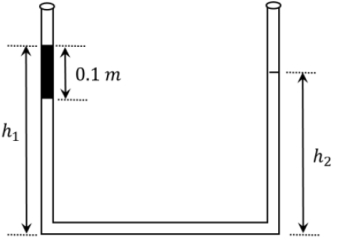
- 7An open-ended U-tube of uniform cross-sectional area contains water (density $10^3 kg m ^{-3}$ ). Initially the water level stands at $0.29 m$ from the bottom in each arm. Kerosene oil (a water-immiscible liquid) of density $800 kg m ^{-3}$ is added to the left arm until its length is $0.1 m$, as shown in the schematic figure below. The ratio $\left(\frac{h_1}{h_2}\right)$ of the heights of the liquid in the two arms is-View Solution
- 8Two non-mixing liquids of densities $\rho $ and $n \rho \,(n > 1)$ are put in a container. The height of each liquid is $h.$ A solid cylinder of length $L$ and density $d$ is put in this container. The cylinder floats with its axis vertical and length $\rho L (\rho < 1)$ in the denser liquid. The density $d$ is equal toView Solution
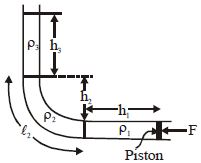
- 9A tube is bent into $L$ shape and kept in a vertical plane. If these three liquids are kept in equilibrium by the piston of area $A$ , the value of $\frac {F}{A}$ isView Solution
- 10A raindrop with radius $R=0.2\, {mm}$ fells from a cloud at a height $h=2000\, {m}$ above the ground. Assume that the drop is spherical throughout its fall and the force of buoyance may be neglected, then the terminal speed attainde by the raindrop is : (In ${ms}^{-1}$)View Solution
[Density of water $f_{{w}}=1000\;{kg} {m}^{-3}$ and density of air $f_{{a}}=1.2\; {kg} {m}^{-3}, {g}=10 \;{m} / {s}^{2}$ Coefficient of viscosity of air $=18 \times 10^{-5} \;{Nsm}^{-2}$ ]
