The specific heat capacity of a metal at low temperature $(T)$ is given as $C_p=32\left(\frac{ T }{400}\right)^{3}\;kJ\,k ^{-1}\, kg ^{-1}$. A $100\; g$ vessel of this metal is to be cooled from $20 \;K$ to $4\; K$ by a special refrigerator operating at room temperature $27^\circ c$). The amount of work required to cool the vessel is
AIEEE 2011, Diffcult
Specific heat at low is
$C_{P}=32\left(\frac{T}{400}\right)^{3}$
$Q=\int m \cdot c . d T=\int_{20}^{4} \frac{100}{1000} \times 32\left(\frac{T}{400}\right)^{3} d T$
$=\frac{32}{10} \times \frac{1}{(400)^{3}}\left(\frac{T^{4}}{4}\right)^{3}$
$=\frac{32}{10 \times(400)^{3}} \times \frac{1}{4}\left(20^{4}-4^{4}\right)$
$=\frac{32}{10 \times(400)^{3}} \times \frac{1}{4} \times(160000-256)$
$=0.002 W$
$\beta=\frac{T_{2}}{T_{1}-T_{2}}=\frac{Q_{3}}{W}$
$\Rightarrow \frac{20}{300-20}=\frac{0.2}{W} \Rightarrow W=0.028 kJ$
$\Rightarrow \frac{4}{300-4}=\frac{0.002}{W} \Rightarrow W=0.0148 kJ$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas at ${27^o}C$ is compressed adiabatically to $\frac{8}{{27}}$ of its original volume. If $\gamma = \frac{5}{3}$, then the rise in temperature is........ $K$View Solution
- 2When $1\, gm$ of water at ${0^o}C$ and $1 \times {10^5}\;N/{m^2}$ pressure is converted into ice of volume $1.091\;c{m^2}$, the external work done will beView Solution
- 3A Car not engine whose low temperate reservoir is at $7\,^oC$ has an efficiency of $50\%$ . It is desired to increase the efficiency to $70\%$ . By how many degrees should the temperature of the high temperature reservoir be increased .... $K$View Solution
- 4An ideal gas heat engine operates in a Carnot cycle between $227^o C$ and $127^o C$. It absorbs $6\,kcal$ at the higher temperature. The amount of heat (in $kcal$) converted into work is equal toView Solution
- 5Jet aircrafts fly at altitudes above $30000 \,ft$, where the air is very cold at $-40^{\circ} C$ and the pressure is $0.28 \,atm$. The cabin is maintained at $1 \,atm$ pressure by means of a compressor which exchanges air from outside adiabatically. In order to have a comfortable cabin temperature of $25^{\circ} C$, we will require in additionView Solution
- 6The graph of pressure $(P)$ and volume $(V)$ according to $PV^n = C$, here $n = 1.4$View Solution
- 7A shown in the figure, the amount of heat absorbed along the path $ABC$ is $90\,J$ and the amount of work done by the system is $30\,J$ . If the amount of work done along the path $ADC$ is $20\,J$ , the amount of heat absorbed will be .... $J$View Solution
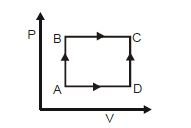
- 8For the given cyclic process $CAB$ as shown for a gas, the work done is ..... $J$View Solution
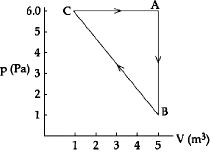
- 9In an isochoric process if ${T_1} = {27^o}C$ and ${T_2} = {127^o}C,$ then ${P_1}/{P_2}$ will be equal toView Solution
- 10During an experiment, an ideal gas is found to obey a condition $\frac{{{P^2}}}{\rho }$ = constant [$\rho =$ density of the gas]. The gas is initially at temperature $T,$ pressure $P$ and density $\rho$ . The gas expands such that density changes to $\rho/2$View Solution
