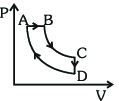
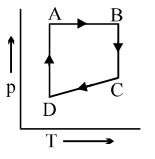
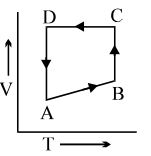
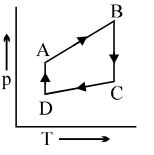
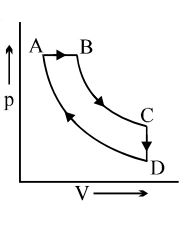
A cyclic process $ABCD$ is shown in the $p-V$ diagram. Which of the following curves represents the same process if $BC \& DA$ are isothermal processes

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A Carnot's heat engine works between the temperatures $427^{\circ} C$ and $27^{\circ} C$. $...........\,kcal / s$ amount of heat should it consume per second to deliver mechanical work at the rate of $1.0\,kW$View Solution
- 2$V\, = \,K\,{\left( {\frac{P}{T}} \right)^{0.33}}$ where $k$ is constant. It is an,View Solution
- 3In case of an adiabatic process the correct relation in terms of pressure $p$ and density $\rho $ of a gas isView Solution
- 4The heat energy required to raise the temperature of $5\,moles$ of an ideal gas to $5\,K$ at constant pressure is $600\,J$ . How much heat (in $J$ ) is required to raise the same mass of the same gas to $5\,K$ at constant volume ? (Take $R = 8.3\,J/mole-^oK$ )View Solution
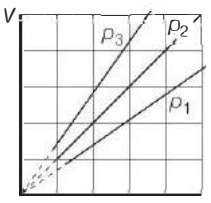
- 5View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
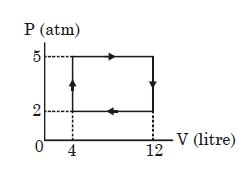
- 6View SolutionAn ideal gas undergoes a cyclic process as shown in diagram. The net work done by the gas in the cycle is
- 7An ideal gas is expanded adiabatically at an initial temperature of $300 K$ so that its volume is doubled. The final temperature of the hydrogen gas is $(\gamma = 1.40)$View Solution
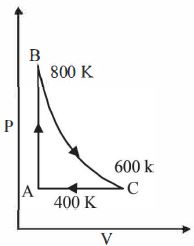
- 8One mole of a diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\ K, 800\ K $ and $600\ K$ respectively. Choose the correct statementView Solution
- 9Consider a carnot's cycle operating between $T_1 = 500\,K$ and $T_2 = 300\,K$ producing $1\,kJ$ of mechanical work per cycle. Find the heat transferred to the engine by the reservoirs .... $J$View Solution
- 10A cyclic process $ABCDA$ is shown in the $P-V$ diagram. Which of the following curves represent the same processView Solution