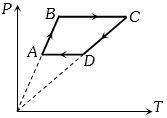
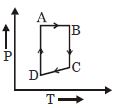
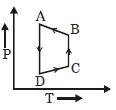
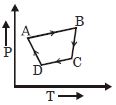
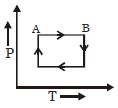
A cyclic process $ABCD$ is shown in the given $P-V$ diagram. $P-T$ diagram that represents the same process is

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionA gas undergoes a change at constant temperature. Which of the following quantities remain fixed?
- 2View SolutionWork done on or by a gas, in general depends upon the
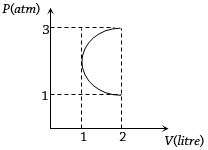
- 3In the $P-V$ diagram shown in figure $ABC$ is a semicircle. The work done in the process $ABC$ isView Solution
- 4View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
- 5A system performs work $\Delta W$ when an amount of heat is$\Delta Q$ added to the system, the corresponding change in the internal energy is $\Delta U$. A unique function of the initial and final states (irrespective of the mode of change) isView Solution
- 6View SolutionWhich relation is correct for isometric process
- 7$5.6$ liter of helium gas at $STP$ is adiabatically compressed to $0.7$ liter. Taking the initial temperature to be $T _1$, the work done in the process isView Solution
- 8View SolutionWhich of the following is incorrect regarding the first law of thermodynamics
- 9$\Delta U + \Delta W = 0$ is valid forView Solution
- 10Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$View Solution