In the $P-V$ diagram shown in figure $ABC$ is a semicircle. The work done in the process $ABC$ is
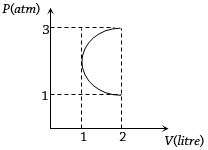

Easy
(b) ${W_{AB}}$ is negative (volume is decreasing) and
${W_{BC}}$ is positive (volume is increasing) and
since, $\left| {\,{W_{BC}}} \right|\, > \,\left| {\,{W_{AB}}} \right|$
$\therefore $ net work done is positive and area between semicircle which is equal to $\frac{\pi }{2}atm - lt.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An ideal gas is expanding such that $\mathrm{PT}^2=$ constant. The coefficient of volume expansion of the gas isView Solution
- 2View SolutionWhich relation is correct for isometric process
- 3View SolutionA given system undergoes a change in which the work done by the system equals the decrease in its internal energy. The system must have undergone an
- 4A Carnot engine working between $300\,K$ and $600\,K$ has work output of $800\, J$ per cycle. What is amount of heat energy supplied to the engine from source per cycle...... $J/cycle$View Solution
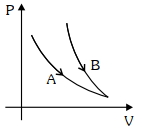
- 5Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
- 6A Carnot engine takes $6000 \,cal$ of heat from a reservoir at $627^{\circ} C$ and gives it to a sink at $27^{\circ} C$. The work done by the engine is ......... $kcal$View Solution
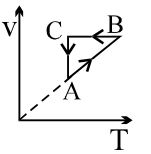
- 7View SolutionAn ideal gas undergoes a thermodynamics cycle as shown in figure. Which of the following graphs represents the same cycle?
- 8If during an adiabatic process the pressure of mixture of gases is found to be proportional to square of its absolute temperature. The ratio of $C_p / C_v$ for mixture of gases is .........View Solution
- 9The efficiency of Carnot engine when source temperature is $T_1$ and sink temperature is $T_2$ will beView Solution
- 10The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
