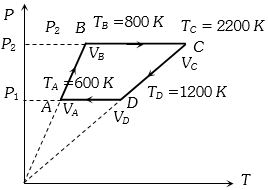
Six moles of an ideal gas perfomrs a cycle shown in figure. If the temperature are $T_A = 600 K, T_B = 800 K, T_C = 2200 K \,and\, T_D = 1200 K$, the work done per cycle is ..... $kJ$

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A gas expands $0.25{m^3}$ at constant pressure ${10^3}N/{m^2}$, the work done isView Solution
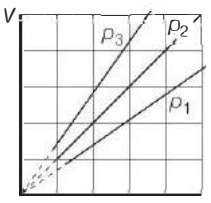
- 2View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
- 3A sample of an ideal gas undergoes an isothermal expansion. If $d Q, d U$ and $d W$ represent the amount of heat supplied, the change in internal energy and the work done respectively, thenView Solution
- 4$1 \,\,kg$ of a gas does $20\,\, kJ$ of work and receives $16 \,\,kJ$ of heat when it is expanded between two states. $A$ second kind of expansion can be found between the initial and final state which requires a heat input of $9\,\, kJ$. The work done by the gas in the second expansion is ....... $kJ$View Solution
- 5View SolutionFor an ideal gas, in an isothermal process
- 6A diatomic ideal gas undergoes a thermodynamic change according to the $P-V$ diagram shown in the figure. The heat given the gas in $AB$View Solution
- 7For an ideal heat engine, the temperature of the source is $127\,^{\circ} C$. In order to have $60\, \%$ efficiency the temperature of the sink should be $........\,{ }^{\circ} C$. (Round off to the Nearest Integer)View Solution
- 8A thermodynamic system is taken through cyclic process. The total work done in the process is $.........\,J$View Solution
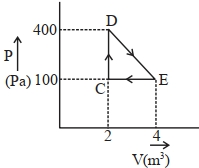
- 9View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
- 10View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is