$\gamma=\frac{5}{3}(F \text { or monoa } \rightarrow \text { micgas })$
The number of moles of gas is $n=\frac{5.6 l}{22.4 l}=\frac{1}{4}$
Finally (after adiabatic compression)
$V_2=0.7 l$
For adiabatic compression $T_1 V_1^{\gamma-1}=T_2 V_2^{\gamma-1}$
$\therefore T_2=T_1\left(\frac{V_1}{V_2}\right)^{\gamma-1}=T_1\left(\frac{5.6}{0.7}\right)^{\frac{5}{3}-1}=T_1(8)^{2 / 3}$
$=4 T_1$
We know that work done in adiabatic process is
$W=\frac{n R \Delta T}{\gamma-1}=\frac{9}{8} R T_1$
Download our appand get started for free
Similar Questions
- 1A Carnot engine absorbs $1000\,J$ of heat energy from a reservoir at $127\,^oC$ and rejects $600\,J$ of heat energy during each cycle. The efficiency of engine and temperature of sink will beView Solution
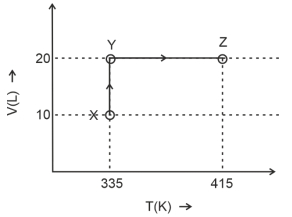
- 2Consider the following volume-temperature $( V - T )$ diagram for the expansion of $5$ moles of an ideal monoatomic gas.View Solution
Considering only $P-V$ work is involved, the total change in enthalpy (in Joule) for the transformation of state in the sequence $X \rightarrow Y \rightarrow Z$ is $\qquad$
[Use the given data: Molar heat capacity of the gas for the given temperature range, $C _{ v , m }=12 J K ^{-1} mol ^{-1}$ and gas constant, $R =8.3 J K ^{-1} mol ^{-1}$ ]
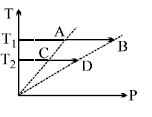
- 3On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
- 4The specific heat at constant pressure of a real gas obeying $\mathrm{PV}^2=\mathrm{RT}$ equation is :View Solution
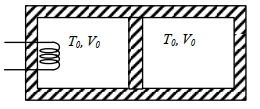
- 5A thermally insulating cylinder has a thermally insulating and frictionless movable partition in the middle, as shown in the figure below. On each side of the partition, there is one mole of an ideal gas, with specific heat at constant volume, $C_v=2 R$. Here, $R$ is the gas constant. Initially, each side has a volume $V_0$ and temperature $T_0$. The left side has an electric heater, which is turned on at very low power to transfer heat $Q$ to the gas on the left side. As a result the partition moves slowly towards the right reducing the right side volume to $V_0 / 2$. Consequently, the gas temperatures on the left and the right sides become $T_L$ and $T_R$, respectively. Ignore the changes in the temperatures of the cylinder, heater and the partition.View Solution
($1$) The value of $\frac{T_R}{T_0}$ is
$(A)$ $\sqrt{2}$ $(B)$ $\sqrt{3}$ $(C)$ $2$ $(D)$ $3$
($2$) The value of $\frac{Q}{R T_0}$ is
$(A)$ $4(2 \sqrt{2}+1)$ $(B)$ $4(2 \sqrt{2}-1)$ $(C)$ $(5 \sqrt{2}+1)$ $(D)$ $(5 \sqrt{2}-1)$
Give the answer or qution ($1$) and ($2$)
- 6During an adiabatic process, if the pressure of a gas is found to be proportional to the cube of its absolute temperature, then the ratio of $\frac{C_p}{C_V}$ for the gas is:View Solution
- 7A Carnot engine operates between two reservoirs of temperatures $900\; \mathrm{K}$ and $300 \;\mathrm{K}$ The engine performs $1200\; \mathrm{J}$ of work per cycle. The heat energy (in $\mathrm{J}$ ) delivered by the engine to the low temperature reservoir, in a cycle. isView Solution
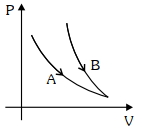
- 8Choose the correct statement for processes $A$ & $B$ shown in figure.View Solution
- 9A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 10A gas ($\gamma = 1.3)$ is enclosed in an insulated vessel fitted with insulating piston at a pressure of ${10^5}\,N/{m^2}$. On suddenly pressing the piston the volume is reduced to half the initial volume. The final pressure of the gas isView Solution
