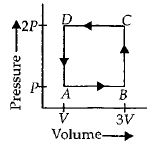
A thermodynamic system is taken through the cycle $ABCD$ as shown in figure. Heat rejected by the gas during the cycle is

AIPMT 2012, Easy
In a cyclic process,
$\Delta U = 0$
In a cyclic process work done is equal to the area under the cycle and is positive if the cycle is clockwise and negative if anticlockwiase.
$\therefore \,\,\,\Delta W = - Area\,of\,rectangle\,ABCD = - P\left( {2V} \right)$
$ = - 2PV$
According to first law of thermodynamics
$\Delta Q = \Delta u + \Delta W\,or\,\Delta Q = \Delta W\,\,\left( {As\,\Delta u = 0} \right)$
$i.e.,$ heat supplied to the system is equal to the work done
So heat absorbed,$\Delta Q = \Delta W = - 2PV$
$\therefore $ Heat rejected by the gas $ = 2PV$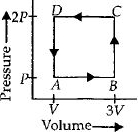
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic gas $\left( {\gamma = \frac{5}{3}} \right)$ is suddenly compressed to $\frac{1}{8}$ of its original volume, then the pressure of gas will change to how many times the initial pressure?View Solution
- 2A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 3A monatomic gas at pressure $P_1$ and volume $V_1$ is compressed adiabatically to ${\frac{1}{8}}^{th}$ of its original volume. What is the final pressure of the gas is ........ $P_1$?View Solution
- 4The process $AB$ is shown in the diagram. As the gas is taken from $A$ to $B$, its temperatureView Solution
- 5Which of the following graphs correctly represents the variation of $\beta = - (dV/dP)/V$ with $P$ for an ideal gas at constant temperatureView Solution
- 6View SolutionThe process in which no heat enters or leaves the system is termed as
- 7On a $TP$ diagram, two moles of ideal gas perform process $AB$ and $CD$. If the work done by the gas in the process $AB$ is two times the work done in the process $CD$ then what is the value of $T_1/T_2$?View Solution
- 8$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 9View SolutionThe adiabatic Bulk modulus of a perfect gas at pressure is given by
- 10Following figure shows $P-T$ graph for four processes $A, B, C$ and $D$. Select the correct alternative.View Solution