Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
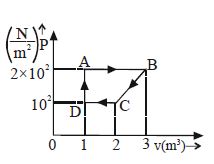
- 1An ideal gas is taken through the cycle $A → B → C → A$, as shown in the figure. If the net heat supplied to the gas in the cycle is $5 \ J$, the work done by the gas in the process $C → A$ is ....... $J$View Solution
- 2$110\; J$ of heat is added to a gaseous system, whose internal energy change is $40\; J$, then the amount of external work done is ........ $J$View Solution
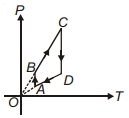
- 3A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
- 4The isothermal Bulk modulus of an ideal gas at pressure $P$ isView Solution
- 5View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
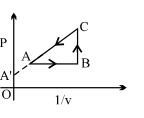
- 6Four curves $A, B, C$ and $D$ are drawn in the adjoining figure for a given amount of gas. The curves which represent adiabatic and isothermal changes areView Solution
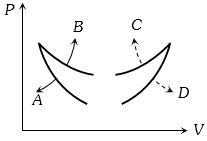
- 7A Carnot engine works between $27^{\circ} C$ and $127^{\circ} C$. Heat supplied by the source is $500\, J$. then heat ejected to the sink is (in $J$)View Solution
- 8$Assertion :$ Adiabatic expansion is always accompanied by fall in temperature.View Solution
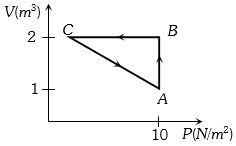
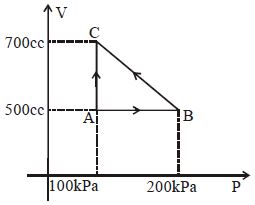
$Reason :$ In adiabatic process, volume is inversely proportional to temperature. - 9A gas taken through cyclic process $ABCA$ is shown in figure. If $2.4\,cal.$ of heat is given in the process, what is value of $J$ ...... $J/cal$ (mechanical equivalent of heat)?View Solution
- 10View SolutionWhen heat is given to a gas in an isothermal change, the result will be