When heat is given to a gas in an isothermal change, the result will be
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Heat energy of $735\,J$ is given to a diatomic gas allowing the gas to expand at constant pressure. Each gas molecule rotates around an internal axis but do not oscillate. The increase in the internal energy of the gas will be $..........\,J$View Solution
- 2A $P-T$ graph is shown for a cyclic process. Select correct statement regarding thisView Solution
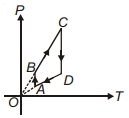
- 3View SolutionA reversible engine and an irreversible engine are working between the same temperatures. The efficiency of the ...........
- 4View SolutionThe work done in an adiabatic change in a gas depends only on
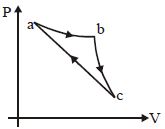
- 5In the $P-V$ diagram shown, the gas does $5\, J$ of work in isothermal process $ab$ and $4\,J$ in adiabatic process $bc$. .... $J$ will be the change in internal energy of the gas in straight path $c$ to $a$ ?View Solution
- 6A Carnot engine whose sink is at $300 \,K$ has an efficiency of $50 \%$. By how much should the temperature of source be increased so as the efficiency becomes $70 \%$ is ............ $K$View Solution
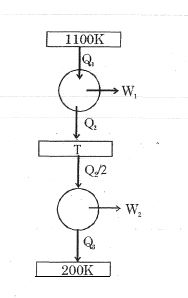
- 7Consider the given series combination of carnot cycles. If $W_1 = W_2$ then the value of $T$ is ...... $K$ (all temperatures are maintained at their respective values)View Solution
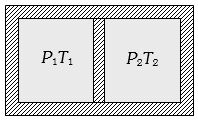
- 8Following figure shows on adiabatic cylindrical container of volume ${V_0}$ divided by an adiabatic smooth piston (area of cross-section = $A$ ) in two equal parts. An ideal gas $({C_P}/{C_V} = \gamma )$ is at pressure $P_1$ and temperature $T_1$ in left part and gas at pressure $P_2$ and temperature $T_2$ in right part. The piston is slowly displaced and released at a position where it can stay in equilibrium. The final pressure of the two parts will be (Suppose $ x$ = displacement of the piston)View Solution
- 9View SolutionBy opening the door of a refrigerator placed inside a room you
- 10View SolutionIf an ideal gas is compressed isothermally then
