The isothermal Bulk modulus of an ideal gas at pressure $P$ is
IIT 1998, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionThe first law of thermodynamics is concerned with the conservation of
- 2Two moles of an ideal monoatomic gas at ${27^o}C$ occupies a volume of $V.$ If the gas is expanded adiabatically to the volume $2V,$ then the work done by the gas will be ....... $J$ $[\gamma = 5/3,\,R = 8.31J/mol\,K]$View Solution
- 3In a thermodynamic process two moles of a monatomic ideal gas obeys $P \propto V^{-2}$. If temperature of the gas increases from $300 \,K$ to $400 \,K$, then find work done by the gas .........$R$ (where $R=$ universal gas constant).View Solution
- 4View SolutionHeat is not being exchanged in a body. If its internal energy is increased, then
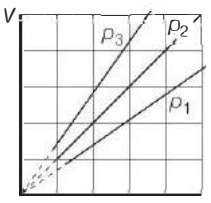
- 5View SolutionThe state of an ideal gas was changed isobarically. The graph depicts three such isobaric lines. Which of the following is true about the pressures of the gas?
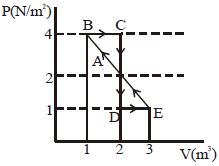
- 6One mole of a monoatomic gas is carried along process $ABCDEA$ as shown in diagram. Find the net work done by gas ......... $J$View Solution
- 7View SolutionThe first law of thermodynamics is concerned with the conservation of
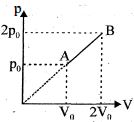
- 8One mole of an ideal gas at $27^{\circ} {C}$ is taken from ${A}$ to ${B}$ as shown in the given ${PV}$ indicator diagram. The work done by the system will be $......\times 10^{-1} \,{J}$View Solution
[Given : $R=8.3\, {J} /\,mole\,{K}, \ln 2=0.6931$ ] (Round off to the nearest integer)
- 9View SolutionA cycle tyre bursts suddenly. This represents an
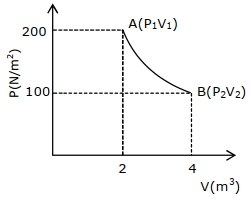
- 10An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution