A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)
Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1View SolutionCofficient of performance of refigerator is
- 2View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
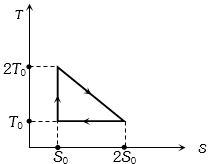
- 3View SolutionIn Carnot engine the work done by working substance is equivalent to
- 4View SolutionIf a cylinder containing a gas at high pressure explodes, the gas undergoes
- 5In an adiabatic process, the state of a gas is changed from ${P_1},{V_1},{T_1} $ to ${P_2},{V_2},{T_2}$. Which of the following relation is correctView Solution
- 6View SolutionWhich relation is correct for isometric process
- 7A student records $\Delta Q, \Delta U \& \Delta W$ for a thermodynamic cycle $A$ $\rightarrow$ $B \rightarrow C \rightarrow A$. Certain entries are missing. Find correct entry in following options.View Solution
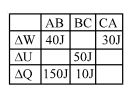
- 8View SolutionA container that suits the occurrence of an isothermal process should be made of
- 9The coefficient of performance of a Carnot refrigerator working between ${30^o}C$ and ${0^o}C$ isView Solution
- 10A heat engine operates with the cold reservoir at temperature $324 K$. The minimum temperature of the hot reservoir, if the heat engine takes $300 \; J$ heat from the hot reservoir and delivers $180 \; J$ heat to the cold reservoir per cycle, is $\dots \; K .$View Solution
