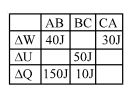
A student records $\Delta Q, \Delta U \& \Delta W$ for a thermodynamic cycle $A$ $\rightarrow$ $B \rightarrow C \rightarrow A$. Certain entries are missing. Find correct entry in following options.

Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If $Q , E$ and $W$ denote respectively the heat added, change in internal energy and the work done by a closed cycle process, thenView Solution
- 2A reversible heat engine converts one-fourth of the heat input into work. When the temperature of the sink is reduced by $52\, K$, its efficiency is doubled. The temperature in Kelvin of the source will be ...... .View Solution
- 3In an $H_2$ gas process, $PV^2 =$ constant. The ratio of work done by gas to change in its internal energy isView Solution
- 4One gm mol of a diatomic gas $(\gamma = 1.4)$ is compressed adiabatically so that its temperature rises from ${27^o}C$ to ${127^o}C$. The work done will beView Solution
- 5$Assertion :$ The heat supplied to a system is always equal to the increase in its internal energy.View Solution
$Reason :$ When a system changes from one thermal equilibrium to another, some heat is absorbed by it. - 6A gas is suddenly compressed to one fourth of its original volume. What will be its final pressure, if its initial pressure is $P$View Solution
- 7View SolutionTwo gases have the same initial pressure, volume and temperatue. They expand to the same final volume, one adiabatically and the other isothermally, if the two gases are compressed to the same final volume
- 8View SolutionPressure versus temperature graph of an ideal gas is shown in figure
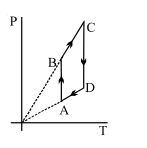
- 9A gas for which $\gamma = 1.5$ is suddenly compressed to $\frac{1}{4}$ th of the initial volume. Then the ratio of the final to the initial pressure isView Solution
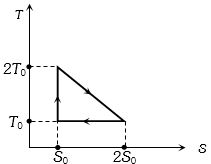
- 10View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is