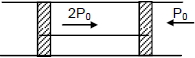
A cylindrical tube of cross-sectional area $A$ has two air tight frictionless pistons at its two ends. The pistons are tied with a straight two ends. The pistons are tied with a straight piece of metallic wire. The tube contains a gas at atmospheric pressure $P_0$ and temperature $T_0$. If temperature of the gas is doubled then the tension in the wire is

Advanced
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1If an ideal gas has volume $V$ at $27°C$ and it is heated at a constant pressure so that its volume becomes $1.5V.$ Then the value of final temperature will be ....... $^oC$View Solution
- 2A container with insulating walls is divided into two equal parts by a partition fitted with a valve. One part is filled with an ideal gas at a pressure $P$ and temperature $T$, whereas the other part is completely evacuated. If the valve is suddenly opened, the pressure and temperature of the gas will beView Solution
- 3The temperature at which root mean square speed of a gas will be half of its value at $0\,^oC$ is ....... $^oC$View Solution
- 4A flask contains hydrogen and oxygen in the ratio of $2: 1$ by mass at temperature $27^{\circ} C$. The ratio of average kinetic energy per molecule of hydrogen and oxygen respectively is :View Solution
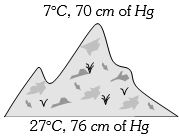
- 5At the top of a mountain a thermometer reads $7°C$ and a barometer reads $70\, cm$ of $Hg.$ At the bottom of the mountain these read $27°C$ and $76 \,cm$ of $Hg$ respectively. Comparison of density of air at the top with that of bottom isView Solution
- 6A gas at ${27}^\circ C$ temperature and $30$ atmospheric pressure is allowed to expand to the atmospheric pressure. If the volume becomes $10$ times its initial volume, then the final temperature becomes ...... $^oC$View Solution
- 7Volume-temperature graph at atmospheric pressure for a monoatomic gas $(V\,{\rm{in }}{m^3},\,\,T\,{\rm{in}}{{\rm{ }}^{\rm{o}}}C)$ isView Solution
- 8The $r.m.s.$ speed of the molecules of a gas at a pressure ${10^5}$ $Pa$ and temperature $0^\circ C$ is $0.5km\;{\sec ^{ - 1}}.$ If the pressure is kept constant but temperature is raised to $819°C,$ the velocity will become ........ $km\,sec^{-1}$View Solution
- 9The internal energy $(U),$ pressure $(P)$ and volume $( V )$ of an ideal gas are related as $U =$ $3 P V+4$. The gas is :-View Solution
- 10View SolutionA sample contains mixture of helium and oxygen gas. The ratio of root mean square speed of helium and oxygen in the sample, is: