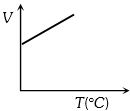
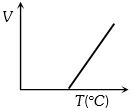
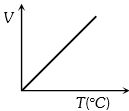
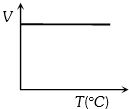
Volume-temperature graph at atmospheric pressure for a monoatomic gas $(V\,{\rm{in }}{m^3},\,\,T\,{\rm{in}}{{\rm{ }}^{\rm{o}}}C)$ is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A vertical closed cylinder is separated into two parts by a frictionless piston of mass $m$ and of negligible thickness. The piston is free to move along the length of the cylinder .The length of the cylinder above the piston is $l_1,$ and that below the piston is $l_2,$ such that $l_1 > l_2.$ Each part of the cylinder contains $n$ moles of an ideal gas at equal temperature $T.$ If the piston is stationary, its mass, $m,$ will be given by: ( $R$ is universal gas constant and $g$ is the acceleration due to gravity)View Solution
- 2View SolutionFor ideal gas, which statement is not true
- 3If $r.m.s.$ velocity of a gas is ${V_{rms}} = 1840m/s$ and its density $\rho = 8.99 \times {10^{ - 2}}kg/{m^3},$ the pressure of the gas will beView Solution
- 4One mole of an ideal gas passes through a process where pressure and volume obey the relation $P\, = {P_0}\,\left[ {1 - \frac{1}{2}{{\left( {\frac{{{V_0}}}{V}} \right)}^2}} \right]$. Here $P_0$ and $V_0$ are constants. Calculate the change in the temperature of the gas if its volume change from $V_0$ to $2V_0$View Solution
- 5In the absence of intermolecular force of attraction, the observed pressure $P$ will beView Solution
- 6Match Column $- I$ and Column $- II$ and choose the correct match from the given choices.View Solution
Column $- I$ Column $- II$ $(A)$ Root mean square speed of gas molecules $(P)$ $\frac{1}{3} \mathrm{n} m \bar{v}^{2}$ $(B)$ Pressure exerted by ideal gas $(Q)$ $\sqrt{\frac{3 \mathrm{RT}}{\mathrm{M}}}$ $(C)$ Average kinetic energy of a molecule $(R)$ $\frac{5}{2} \mathrm{RT}$ $(D)$ Total internal energy of $1$ mole of a diatomic gas $(S)$ $\frac{3}{2} \mathrm{k}_{\mathrm{B}} \mathrm{T}$ - 7The speeds of $5$ molecules of a gas (in arbitrary units) are as follows : $2, 3, 4, 5, 6.$ The root mean square speed for these molecules isView Solution
- 8A pressure cooker contains air at $1$ atm and $30^o C$. If the safety value of the cooler blows when the inside pressure $ \ge 3$ atm, then the maximum temperature of the air, inside the cooker can be .... $^oC$View Solution
- 9View SolutionWhat will be the effect on the root mean square velocity of oxygen molecules if the temperature is doubled and oxygen molecule dissociates into atomic oxygen?
- 10The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution