A diatomic ideal gas is used in a Carnot engine as the working substance. If during the adiabatic expansion part of the cycle the volume of the gas increases from $V$ to $32\ V$, the efficiency of the engine is
AIEEE 2010, Medium
${T_1}{V^{\gamma - 1}} = {T_2}{\left( {32V} \right)^{\gamma - 1}} \Rightarrow {T_1} = {\left( {32} \right)^{\gamma - 1}}.{T_2}$
$For\,diatomic\,gas,\,\gamma = \frac{7}{5}\,\,\,\,\,\,\therefore \gamma - 1 = \frac{2}{5}$
$\therefore {T_1} = {\left( {32} \right)^{\frac{2}{5}}}.{T_2} \Rightarrow {T_1} = 4{T_2}$
$Now,\,efficiency = 1 - \frac{{{T_2}}}{{{T_1}}} = 1 - \frac{1}{4} = \frac{3}{4} = 0.75.$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
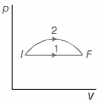
- 1An ideal gas undergoes change in its state from the initial state $I$ to the final state $F$ via two possible paths as shown below. Then,View Solution
- 2An engineer claims to have made an engine delivering $10 kW$ power with fuel consumption of $1 g/sec$. The calorific value of the fuel is $2 kcal/g$. Is the claim of the engineerView Solution
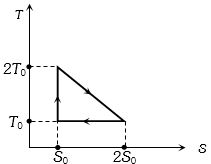
- 3View SolutionThe temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
- 4View SolutionIn isothermic process, which statement is wrong
- 5View SolutionThe door of a working refrigerator is left open in a well insulated room. The temperature of air in the room will
- 6View SolutionIrreversible process is
- 7For an ideal gas, the internal energy is given by $U=5 p V / 2+C$, where $C$ is a constant. The equation of the adiabats in the $p V$-plane will beView Solution
- 8A cylinder of mass $1\,kg$ is given heat of $20000\, J$ at atmospheric pressure. If initially temperature of cylinder is $20\,^oC$, then work done by the cylinder will be .......$J$ (Given that Specific heat of cylinder $= 400 \,J\, kg^{-1}$, Coefficient of volume expansion $= 9 \times {10^{-5}}\,^o C^{-1}$, Atmospheric pressure $= 10^5 \,N/m^2$ and density of cylinder $9000\,kg/m^3$)View Solution
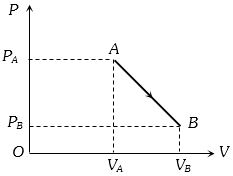
- 9An ideal gas is taken from point $A$ to the point $B,$ as shown in the $P-V$ diagram, keeping the temperature constant. The work done in the process isView Solution
- 10One mole of an ideal gas is taken through an adiabatic process where the temperature rises from $27^{\circ} {C}$ to $37^{\circ} {C}$. If the ideal gas is composed of polyatomic molecule that has $4$ vibrational modes which of the following is true?View Solution
