The temperature-entropy diagram of a reversible engine cycle is given in the figure. Its efficiency is
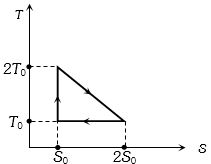

AIEEE 2005, Diffcult
${Q_1} = {T_0}{S_0} + \frac{1}{2}{T_0}{S_0} = \frac{3}{2}{T_0}{S_0}$
${Q_2} = {T_0}{S_0}$ and ${Q_3} = 0$
$\eta = \frac{W}{{{Q_1}}} = \frac{{{Q_1} - {Q_2}}}{{{Q_1}}}$
$ = 1 - \frac{{{Q_2}}}{{{Q_1}}} = 1 - \frac{2}{3} = \frac{1}{3}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1$Assertion :$ In free expansion of an ideal gas, the entropy increases.View Solution
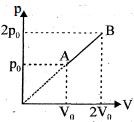
$Reason :$ Entropy increases in all natural processes. - 2An enclosed one mole of a monoatomic gas is taken through a process $A$ to $B$ as shown in figure. The molar heat capacity of the gas for this process isView Solution
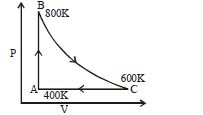
- 3One mole of diatomic ideal gas undergoes a cyclic process $ABC$ as shown in figure. The process $BC$ is adiabatic. The temperatures at $A, B$ and $C$ are $400\,K, 800\,K$ and $600\,K$ respectively. Choose the correct statementView Solution
- 4View SolutionAn enclosed ideal gas is taken through a cycle as shown in the figure. Then
- 5A gas expands $0.25{m^3}$ at constant pressure ${10^3}N/{m^2}$, the work done isView Solution
- 6In a thermodynamic process, pressure of a fixed mass of a gas is changed in such a manner that the gas molecules gives out $20 J$ of heat and $10 J$ of work is done on the gas. If the initial internal energy of the gas was $40 J,$ then the final internal energy will be ........ $J$View Solution
- 7View SolutionFirst law of thermnodynamics is given by
- 8A Carnot's engine working between $400\, K$ and $800\, K$ has a work output of $1200\, J$ per cycle. The amount of heat energy supplied to the engine from the source in each cycle is ........... $J$View Solution
- 9A gas expands with temperature according to the relation $V = k{T^{2/3}}.$ What is the work done when the temperature changes by ${30^o}C$View Solution
- 10Three moles of an ideal monoatomic gas perform a cycle as shown in the figure. The gas temperature in different states are: $T_1 = 400\, K, T_2 = 800\, K, T_3 = 2400\,K$ and $T_4 = 1200\,K.$ The work done by the gas during the cycle is ........ $kJ$View Solution