A flask contains Hydrogen and Argon in the ratio $2: 1$ by mass. The temperature of the mixture is $30^{\circ} C$. The ratio of average kinetic energy per molecule of the two gases ( $K$ argon/ $K$ hydrogen) is: (Given: Atomic Weight of $Ar = 39.9$)
JEE MAIN 2023, Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1In case of hydrogen and oxygen at $N.T.P.$, which of the following quantities is $/$ are the same?View Solution
- 2$Assertion :$ The total translational kinetic energy of all the molecules of a given mass of an ideal gas is $1.5$ times the product of its pressure and its volume.View Solution
$Reason :$ The molecules of a gas collide with each other and the velocities of the molecules change due to the collision. - 3The molar heat capacity in a process of a diatomic gas if it does a work of $\frac{Q}{4}$ when a heat of $Q$ is supplied to it isView Solution
- 4View SolutionGraph of specific heat at constant volume for a monoatomic gas is
- 5In the two vessels of same volume, atomic hydrogen and helium at pressure $1\, atm$ and $2\, atm$ are filled. If temperature of both the samples is same, then average speed of hydrogen atoms $ < {C_H} > $ will be related to that of helium $ < {C_{He}} > $ asView Solution
- 6The molar specific heat of mixture at constant volume, if one mole of $He$ gas is mixed with three moles of $O_2$ gas isView Solution
- 7An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $8.0\times10^{-3}\, m^2$ . Initially the gas is at $300\, K$ and occupies a volume of $2.4\times10^{-3}\, m^3$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by $0.1\, m$. The force constant of the spring is $8000\, N/m$ and the atmospheric pressure is $1.0\times10^5\, N/m^2$ . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater . The heat capacity of the heater coil is also negligible)View Solution
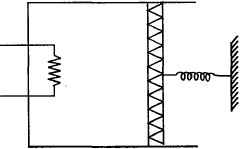
- 8The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?View Solution
- 9At what temperature is the kinetic energy of a gas molecule double that of its value of $27°C$View Solution
- 10One kg of a diatomic gas is at a pressure of $8 \times 10^4$ $N/m^2$ The density of the gas is $4$ $kg/m^3$ What is the energy (in $\times 10^4\; J$) of the gas due to its thermal motion?View Solution
