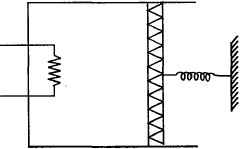
An ideal monoatomic gas is confined in a cylinder by a spring loaded piston of cross section $8.0\times10^{-3}\, m^2$ . Initially the gas is at $300\, K$ and occupies a volume of $2.4\times10^{-3}\, m^3$ and the spring is in its relaxed state as shown in figure. The gas is heated by a small heater until the piston moves out slowly by $0.1\, m$. The force constant of the spring is $8000\, N/m$ and the atmospheric pressure is $1.0\times10^5\, N/m^2$ . The cylinder and the piston are thermally insulated. The piston and the spring are massless and there is no friction between the piston and the cylinder. The final temperature of the gas will be: (Neglect the heat loss through the lead wires of the heater . The heat capacity of the heater coil is also negligible)

JEE MAIN 2014, Diffcult
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Five moles of helium are mixed with two moles of hydrogen to form a mixture. Take molar mass of helium $M_1=4\ g$ and that of hydrogen $M_2=2\ g$ The equivalent degree of freedom $f$ of the mixture isView Solution
- 2A jar has a mixture of hydrogen and oxygen gas in the ratio of $1 : 5$. The ratio of mean kinetic energies of hydrogen and oxygen molecules isView Solution
- 3Five particles have speeds $1, 2, 3, 4, 5 \,\,m/s$. the average velocity of the particles is (in $m/s$)View Solution
- 4The relationship between pressure and the density of a gas expressed by Boyle’s law, $ P = KD$ holds trueView Solution
- 5One mole of an ideal gas requires $207\, J$ heat to raise the temperature by $10 \,K$ when heated at constant pressure. If the same gas is heated at constant volume to raise the temperature by the same $10\, K,$ the heat required is ...... $J$View Solution
(Given the gas constant $R = 8.3J/mol{\rm{ - }}K$)
- 6From the following $V-T$ diagram we can concludeView Solution
- 7A spherical bubble inside water has radius $R$. Take the pressure inside the bubble and the water pressure to be $p_0$. The bubble now gets compressed radially in an adiabatic manner so that its radius becomes $(R-a)$. For $a \ll R$ the magnitude of the work done in the process is given by $\left(4 \pi p_0 R a^2\right) X$, where $X$ is a constant and $\gamma=C_p / C_V=41 / 30$. The value of $X$ is. . . . . .View Solution
- 8In the kinetic theory of gases, which of these statements is/are true ?View Solution
$(i)$ The pressure of a gas is proportional to the mean speed of the molecules.
$(ii)$ The root mean square speed of the molecules is proportional to the pressure.
$(iii)$ The rate of diffusion is proportional to the mean speed of the molecules.
$(iv)$ The mean translational kinetic energy of a gas is proportional to its kelvin temperature.
- 9The molecular weights of $O_2$ and $N_2$ are $32$ and $28$ respectively. At $15°C,$ the pressure of $1 \,gm$ $O_2$ will be the same as that of $1 \,gm$ $N_2$ in the same bottle at the temperature ...... $^oC$View Solution
- 10The temperature at which the kinetic energy of oxygen molecules becomes double than its value at $27^{\circ}\,C$ is $............^{\circ}\,C$View Solution
