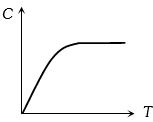
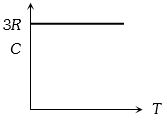
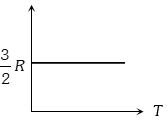
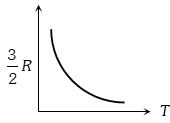
Graph of specific heat at constant volume for a monoatomic gas is
Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An object is placed in a medium of refractive index $3$. An electromagnetic wave of intensity $6 \times 10^8 \mathrm{~W} / \mathrm{m}^2$ falls normally on the object and it is absorbed completely. The radiation pressure on the object would be (speed of light in free space $=3 \times 10^8 \mathrm{~m} / \mathrm{s}$ ):View Solution
- 2A balloon contains $500\,{m^3}$ of helium at $27°C$ and $1$ atmosphere pressure. The volume of the helium at $-3°C$ temperature and $0.5$ atmosphere pressure will be ..... $m^3$View Solution
- 3$Assertion :$ At a given temperature the specific heat of a gas at constant pressure. is always greater than its specific heat at constant volumeView Solution
$Reason :$ When a gas is heated at constant volume some extra heat is needed compared to that at constant pressure for doing work in expansion.
- 4View SolutionA triatomic, diatomic and monatomic gas is supplied same amount of heat at constant pressure, then
- 5Air is pumped into an automobile tube upto a pressure of $200\, kPa$ in the morning when the air temperature is $22°C.$ During the day, temperature rises to $42°C$ and the tube expands by $2\%.$ The pressure of the air in the tube at this temperature, will be approximately ...... $kPa$View Solution
- 6The root mean square speed of molecules of nitrogen gas at $27^{\circ} C$ is approximately$.......m/s$(Given mass of a nitrogen molecule $=4.6 \times 10^{-26}\,kg$ and take Boltzmann constant $k _{ B }=1.4 \times 10^{-23}\,JK ^{-1}$ )View Solution
- 7View SolutionInternal energy of a non-ideal gas depends on ..........
- 8An ideal gas initially at pressure $1$ bar is being compressed from $30 \,m ^{3}$ to $10\, m ^{3}$ volume and its temperature decreases from $320\, K$ to $280\, K$. then find final pressure of gas (in bar)View Solution
- 9The value closest to the thermal velocity of a Helium atom at room temperature $(300\,K)$in $ms^{-1}$ is $[k_B\, = 1 .4\times10^{-23}\,J/K;\, m_{He}\, = 7\times10^{-27}\,kg]$View Solution
- 10Three particles have speeds of $2u$ , $10u$ and $11u$. Which of the following statements is correct?View Solution