The temperature of a gas is $-50^{\circ}\,C$. To what temperature the gas should be heated so that the rms speed is increased by $3$ times?
NEET 2023, Easy
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1At what temperature is the root mean square velocity of gaseous hydrogen molecules is equal to that of oxygen molecules at $47°C$ ..... $K$View Solution
- 2The mean free path of gas molecules depends on $(d =$ molecular diameter$)$View Solution
- 3If an ideal gas has volume $V$ at $27°C$ and it is heated at a constant pressure so that its volume becomes $1.5V.$ Then the value of final temperature will be ....... $^oC$View Solution
- 4View SolutionOn colliding in a closed container the gas molecules
- 5For a gas $\gamma = 7/5.$ The gas may probably beView Solution
- 6View SolutionThat gas cannot be liquified
- 7A hydrogen cylinder is designed to withstand an internal pressure of $100 \,atm$. At $27^{\circ} C$, hydrogen is pumped into the cylinder which exerts a pressure of $20 \,atm$. At what temperature does the danger of explosion first sets in ......... $K$View Solution
- 8The respective speeds of the molecules are $1, 2, 3, 4$ and $5\, km/sec.$ The ratio of their $r.m.s. $ velocity and the average velocity will beView Solution
- 9Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
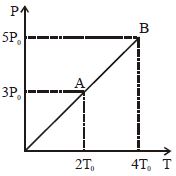
- 10View SolutionSelect the incorrect statement about the specific heats of a gaseous system.
