A gas at $ 27\ ^oC$ has a volume $V$ and pressure $P$. On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ....... $^oC$
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1An air bubble doubles its radius on raising from the bottom of water reservoir to be the surface of water in it. If the atmospheric pressure is equal to $10\, m$ of water, the height of water in the reservoir is ..... $m$View Solution
- 2View SolutionThe effect of temperature on Maxwell's speed distribution is correctly shown by
- 3The root mean square speed of molecules of nitrogen gas at $27^{\circ} C$ is approximately$.......m/s$(Given mass of a nitrogen molecule $=4.6 \times 10^{-26}\,kg$ and take Boltzmann constant $k _{ B }=1.4 \times 10^{-23}\,JK ^{-1}$ )View Solution
- 4Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ isView Solution
- 5View SolutionIn thermal equilibrium, the average velocity of gas molecules is
- 6Air is filled at $60^o C$ in a vessel of open mouth. The vessel is heated to a temperature $T$ so that $1/4^{th}$ part of air escapes. Assuming the volume of the vessel remaining constant, the value of $T$ is ....... $^oC$View Solution
- 7When a gas filled in a closed vessel is heated by raising the temperature by $1^{\circ} C$, its pressure increase by $0.4 \%$. The initial temperature of the gas is ..........$K$View Solution
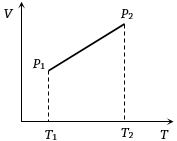
- 8From the following $V-T$ diagram we can concludeView Solution
- 9A gas at $27^o C$ has a volume $V$ and pressure $P.$ On heating its pressure is doubled and volume becomes three times. The resulting temperature of the gas will be ...... $^oC$View Solution
- 10Two identical bodies are made of a material for which the heat capacity increases with temperature. One of these is held at a temperature of $100^{\circ} C$, while the other one is kept at $0^{\circ} C$. If the two are brought into contact, then assuming no heat loss to the environment, the final temperature that they will reach isView Solution
