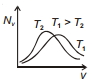
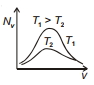
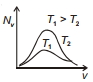
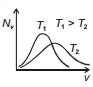
The effect of temperature on Maxwell's speed distribution is correctly shown by
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1A monoatomic gas performs a work of $\frac{Q}{4}$ where $Q$ is the heat supplied to it. The molar heat capaticy of the gas will be $R$ during this transformation. Where $R$ is the gas constant.View Solution
- 2An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume $V_1$ and contains ideal gas at pressure $P_1$ and temperature $T_1$ . The other chamber has volume $V_2$ and contains ideal gas at pressure $P_2$ and temperature $T_2$. If the partition is removed without doing any work on the gas, the final equilibrium temperature of the gas in the container will beView Solution
- 3The lowest pressure (the best Vacuum) that can be created in the laboratory at $27$ degree is $10^{-11} \;{mm}$ of $Hg$. At this pressure, the number of ideal gas molecules per ${cm}^{3}$ will beView Solution
- 4At $0 \;K$ which of the following properties of a gas will be zeroView Solution
- 5View SolutionWhich of the following formulae is wrong
- 6If the root mean square velocity of the molecules of hydrogen at $NTP$ is $1.84\, km/s$. Calculate the root mean square velocity of oxygen molecule at $NTP$, molecular weight of hydrogen and oxygen are $2$ and $32$ respectively ....... $km/sec$View Solution
- 7The graph which represent the variation of mean kinetic energy of molecules with temperature $t°C$ isView Solution
- 8What is the mass of $2$ litres of nitrogen at $22.4$ atmospheric pressure and $273\,K$View Solution
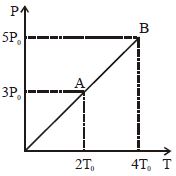
- 9Pressure $Vs$ temperature graph of an ideal gas is shown in figure. Density of the gas at point $A$ is $\rho _0$ , Density at $B$ will beView Solution
- 10Nitrogen gas $N _2$ of mass $28 \,g$ is kept in a vessel at pressure of $10 \,atm$ and temperature $57^{\circ} C$. Due to leakage of $N _2$ gas its pressure falls to $5 \,atm$ and temperature to $27^{\circ} C$. The amount of $N _2$ gas leaked out is .......... $g$View Solution