Equation of gas in terms of pressure $(P),$ absolute temperature $(T)$ and density $(d)$ is
Medium
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Which statements are correct about degrees of freedom?View Solution
$A.$ $A$ molecule with $n$ degrees of freedom has $n^{2}$ different ways of storing energy.
$B.$ Each degree of freedom is associated with $\frac{1}{2} RT$ average energy per mole.
$C.$ $A$ monoatomic gas molecule has $1$ rotational degree of freedom where as diatomic molecule has $2$ rotational degrees of freedom
$D$ $CH _{4}$ has a total to $6$ degrees of freedom Choose the correct answer from the option given below:
- 2The root mean square velocity of a gas molecule of mass $m$ at a given temperature is proportional toView Solution
- 3When one mole of monatomic gas is mixed with one mole of a diatomic gas, then the equivalent value of $\gamma$ for the mixture will be (vibration mode neglected)View Solution
- 4The equation of state corresponding to $8 \,g$ of ${O_2}$ isView Solution
- 5At a given temperature the ratio of $r.m.s.$ velocities of hydrogen molecule and helium atom will beView Solution
- 6If the root mean square velocity of hydrogen molecule at a given temperature and pressure is $2 \mathrm{~km} / \mathrm{s}$, the root mean square velocity of oxygen at the same condition in $\mathrm{km} / \mathrm{s}$ is :View Solution
- 7At constant pressure, the ratio of increase in volume of an ideal gas per degree raise in kelvin temperature to it's original volume is $(T =$ absolute temperature of the gas$)$View Solution
- 8A gas in container $A$ is in thermal equilibrium with another gas in container $B.$ both contain equal masses of the two gases in the respective containers. Which of the following can be trueView Solution
- 9$PV$ versus $T$ graph of equal masses of ${H_2}$, $He$ and ${O_2}$ is shown in fig. Choose the correct alternativeView Solution
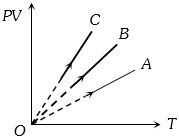
- 10A mixture of ideal gases $7\,\, kg$ of nitrogen and $11 \,\,kg$ of $CO_2$. ThenView Solution
(Take $\gamma$ for nitrogen and $CO_2$ as $1.4$ and $1.3$ respectively)
