A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphere
Medium
(c) $P{V^\gamma } = {\rm{constant}} \Rightarrow \frac{{{P_2}}}{{{P_1}}} = {\left( {\frac{{{V_1}}}{{{V_2}}}} \right)^\gamma }$$ \Rightarrow \frac{{{P_2}}}{1} = {\left( {\frac{{{V_1}}}{{{V_1}/4}}} \right)^{3/2}} = 8$
==> ${P_2} = 8\;atm$.
==> ${P_2} = 8\;atm$.
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Starting with the same initial conditions, an ideal gas expands from volume $V_{1}$ to $V_{2}$ in three different ways. The work done by the gas is $W_{1}$ if the process is purely isothermal. $W _{2}$. if the process is purely adiabatic and $W _{3}$ if the process is purely isobaric. Then, choose the coned optionView Solution
- 2A gas may expand either adiabatically or isothermally. A number of $P-V$ curves are drawn for the two processes over different range of pressure and volume. It will be found thatView Solution
- 3In an adiabatic process $90J$ of work is done on the gas. The change in internal energy of the gas is ....... $J$View Solution
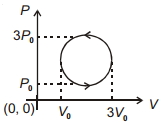
- 4View SolutionWork done in the cyclic process shown in figure is ...........
- 5A motor-car tyre has a pressure of $2\, atm$ at $27\,^oC$. It suddenly burst's. If $\left( {\frac{{{C_p}}}{{{C_v}}}} \right) = 1.4$ for air, find the resulting temperatures (Given $4^{1/7} = 1.219$)View Solution
- 6$100\ g$ of water is heated from $30^o\ C$ to $50^o\ C$ Ignoring the slight expansion of the water, the change in its internal energy is ...... $kJ$ (specific heat of water is $4184\ J/kg/K$)View Solution
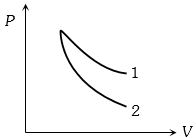
- 7$P-V$ plots for two gases during adiabatic process are shown in the figure. Plots $1$ and $2$ should correspond respectively toView Solution
- 8Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
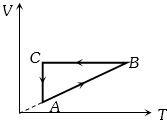
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 9A cyclic process $ABCA$ is shown in the $V-T $ diagram. Process on the $P-V$ diagram isView Solution
- 10A balloon filled with helium $\left(32^{\circ} C \right.$ and $1.7\; atm$.) bursts. Immediately afterwards the expansion of helium can be considered asView Solution
