$W _{2} < W _{1} < W _{3}$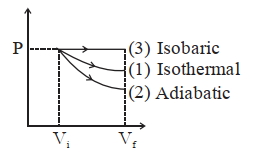
Download our appand get started for free
Similar Questions
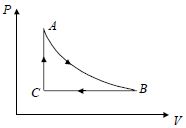
- 1A fixed amount of a gas undergoes a thermodynamic process as shown such that heat interaction along path $B \to C \to A$ is equal to the work done by the gas along path $A \to B \to C$. Then process $A \to B$ is :-View Solution
- 2Air is pumped into a balloon, of initial volume $V$ , until its diameter has doubled. If the atmospheric pressure is $p$ , what is the work done against the atmosphere ?View Solution
- 3$Assertion :$ The Carnot cycle is useful in understanding the performance of heat engines.View Solution
$Reason :$ The Carnot cycle provides a way of determining the maximum possible efficiency achievable with reservoirs of given temperatures. - 4The temperature of food material in refrigerator is $4^{\circ} C$ and temperature of environment is $15^{\circ} C$. If carnot cycle is used in its working gas, then find its carnot efficiency.View Solution
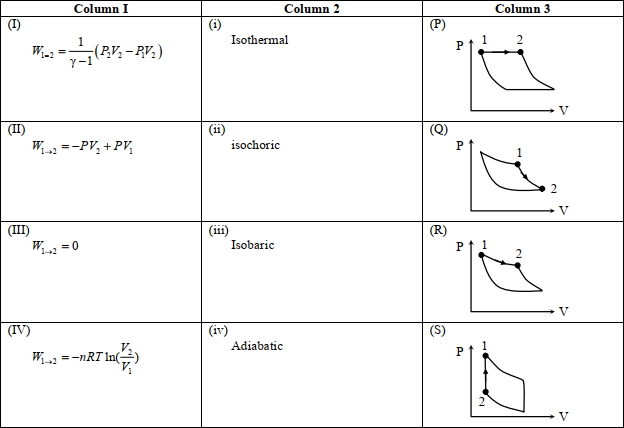
- 5An ideal gas is undergoing a cyclic thermodynamic process in different ways as shown in the corresponding $P$ $V$ diagrams in column $3$ of the table. Consider only the path from state $1$ to $2 . W$ denotes the corresponding work done on the system. The equations and plots in the table have standard notations as used in thermodynamic processes. Here $\gamma$ is the ratio of heat capacities at constant pressure and constant volume. The number of moles in the gas is $n$.View Solution
(image)
($1$) Which of the following options is the only correct representation of a process in which $\Delta U=\Delta Q-P \Delta V$ ?
$[A] (II) (iv) (R)$ $[B] (II) (iii) (P)$ $[C] (II) (iii) (S)$ $[D] (III) (iii) (P)$
($2$) Which one of the following options is the correct combination?
$[A] (III) (ii) (S)$ $[B] (II) (iv) (R)$ $[C] (II) (iv) (P)$ $[D] (IV) (ii) (S)$
($3$) Which one of the following options correctly represents a thermodynamic process that is used as a correction in the determination of the speed of sound in an ideal gas?
$[A] (III) (iv) (R)$ $[B] (I) (ii)$ $(\mathrm{Q})$ $[C] (IV) (ii) (R)$ $[D] (I) (iv) (Q)$
- 6$Assertion :$ When a glass of hot milk is placed in a room and allowed to cool, its entropy decreases.View Solution
$Reason :$ Allowing hot object to cool does not violate the second law of thermodynamics. - 7A gas is suddenly compressed to $1/4$ th of its original volume at normal temperature. The increase in its temperature is ....... $K$ $(\gamma = 1.5)$View Solution
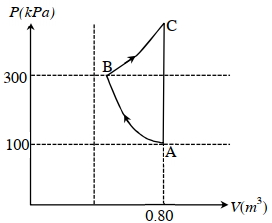
- 8If a gas is taken from $A$ to $C$ through $B$ then heat absorbed by the gas is $8 \,J$. Heat absorbed by the gas in taking it from $A$ to $C$ directly is ............. $J$View Solution
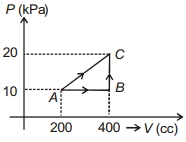
- 9In the given $P$-V diagram, a monoatomic gas $\left(\gamma=\frac{5}{3}\right)$ is first compressed adiabatically from state $A$ to state $B$. Then it expands isothermally from state $B$ to state $C$. [Given: $\left(\frac{1}{3}\right)^{0.6} \simeq 0.5, \ln 2 \simeq 0.7$ ].View Solution
Which of the following statement($s$) is(are) correct?
$(A)$ The magnitude of the total work done in the process $A \rightarrow B \rightarrow C$ is $144 kJ$.
$(B)$ The magnitude of the work done in the process $B \rightarrow C$ is $84 kJ$.
$(C)$ The magnitude of the work done in the process $A \rightarrow B$ is $60 kJ$.
$(D)$ The magnitude of the work done in the process $C \rightarrow A$ is zero.
- 10View SolutionA perfect gas contained in a cylinder is kept in vacuum. If the cylinder suddenly bursts, then the temperature of the gas
