Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the system
| Process | Condition |
| $(I)$ Adiabatic | $(A)\; \Delta W =0$ |
| $(II)$ Isothermal | $(B)\; \Delta Q=0$ |
| $(III)$ Isochoric | $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ |
| $(IV)$ Isobaric | $(D)\; \Delta U =0$ |
JEE MAIN 2020, Medium
$(I)$ Adiabatic process $\Rightarrow \Delta Q=0$ No exchange of heat takes place with surroundings
$(II)$ Isothermal proess $\Rightarrow$ Temperature remains constant $(\Delta T =0)$
$\Delta u =\frac{ F }{2} nR \Delta T \Rightarrow \Delta u =0$
No change in internal energy $[\Delta u =0]$
$(III)$ Isochoric process Volume remains constant
$\Delta V =0$
$W =\int P \cdot d V =0$
Hence work done is zero.
$(IV)$ Isobaric process $\Rightarrow$ Pressure remains constant
$W = P . \Delta V \neq 0$
$\Delta u =\frac{ F }{2} nR \Delta T =\frac{ F }{2}[ P \Delta V ] \neq 0$
$\Delta Q = n C _{ p } \Delta T \neq 0$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
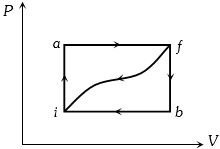
- 1When a system is taken from state $i$ to a state $f$ along path $iaf, \,Q = 50\,J$ and $W = 20J.$ Along path $ibf, \,Q = 35J.$ If $W = - 13J$ for the curved return path $f i, Q$ for this path is ...... $J$View Solution
- 2If the amount of heat given to a system be $35$ joules and the amount of work done by the system be $ - 15$ joules, then the change in the internal energy of the system is .... $joules$View Solution
- 3View SolutionIn the case of diatomic gas, the heat given at constant pressure is that part of energy which is used for the expansion of gas, is
- 4View SolutionAt constant pressure how much fraction of heat supplied to gas is converted into mechanical work?
- 5Match the thermodynamic processes taking place in a system with the correct conditions. In the table: $\Delta Q$ is the heat supplied, $\Delta W$ is the work done and $\Delta U$ is change in internal energy of the systemView Solution
Process Condition $(I)$ Adiabatic $(A)\; \Delta W =0$ $(II)$ Isothermal $(B)\; \Delta Q=0$ $(III)$ Isochoric $(C)\; \Delta U \neq 0, \Delta W \neq 0 \Delta Q \neq 0$ $(IV)$ Isobaric $(D)\; \Delta U =0$ - 6In a mechanical refrigerator, the low temperature coils are at a temperature of $-23°C$ and the compressed gas in the condenser has a temperature of $27°C.$ The theoretical coefficient of performance isView Solution
- 7$5.6$ liter of helium gas at $STP$ is adiabatically compressed to $0.7$ liter. Taking the initial temperature to be $T _1$, the work done in the process isView Solution
- 8Areversible adiabatic path on a $P-V$ diagram for an ideal gas passes through stateAwhere $P=0$.$7\times 10^5 \,\,N/ m^{-2}$ and $v = 0.0049 \,\,m^3$. The ratio of specific heat of the gas is $1.4$. The slope of path at $A$ is :View Solution
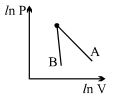
- 9The figure, shows the graph of logarithmic reading of pressure and volume for two ideal gases $A$ and $B$ undergoing adiabatic process. From figure it can be concluded thatView Solution
- 10Two moles of helium gas are taken over the cycle $ABCDA$, as shown in the $P-T$ diagram.The work done on the gas in taking it from $D$ to $A$ isView Solution

