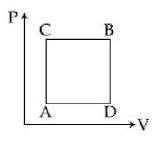
A gas can be taken from $A$ to $B$ via two different processes $ACB$ and $ADB$. When path $ACB$ is used $60\, J$ of heat flows into the system and $30\, J$ of work is done by the system. If path $ADB$ is used work down by the system is $10\, J$. the heat flow into the system in path $ADB$ is ..... $J$

JEE MAIN 2019, Medium
As temperature at point $A$ and $C$ is same.
$\therefore$ Internal energy change will be same.
$\mathrm{Q}-\mathrm{W}=\mathrm{Q}^{\prime}-\mathrm{W}^{\prime}$
$60-30=Q^{\prime}-10$
$Q^{\prime}=40 \mathrm{J}$
Download our appand get started for free
Experience the future of education. Simply download our apps or reach out to us for more information. Let's shape the future of learning together!No signup needed.*
Similar Questions
- 1Two gases of equal mass are in thermal equilibrium. If ${P_a},\,{P_b}$ and ${V_a}$ and ${V_b}$ are their respective pressures and volumes, then which relation is trueView Solution
- 2Two kg of water is converted into steam by boiling at atmospheric pressure. The volume changes from $2 \times {10^{ - 3}}\,{m^3}$ to $3.34{m^3}.$ The work done by the system is about ....... $kJ$View Solution
- 3View SolutionCan two isothermal curves cut each other
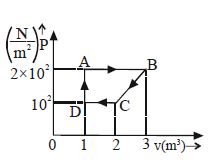
- 4A cyclic process is shown in figure. Work done during isobaric expansion is ...... $J$View Solution
- 5One mole of an ideal gas at an initial temperature of $T\, K$ does $6 R$ joules of work adiabatically. If the ratio of specific heats of this gas at constant pressure and at constant volume is $5/3$, the final temperature of gas will beView Solution
- 6Three moles of an ideal gas $\left( {{C_P} = \frac{7}{2}R} \right)$ at pressure ${P_A}$ and temperature ${T_A}0$ is isothermally expanded to twice its initial volume. It is then compressed at constant pressure to its original volume. Finally the gas is compressed at constant volume to its original pressure ${P_A}.$ The correct $P-V$ and $P-T$ diagrams indicating the process areView Solution
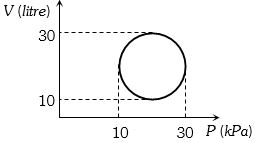
- 7View SolutionHeat energy absorbed by a system in going through a cyclic process shown in figure is
- 8View SolutionAn ideal diatomic gas is heated at constant pressure. The ratio of the work done to the heat supplied is
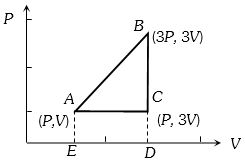
- 9An ideal gas is taken around $ABCA$ as shown in the above $P-V$ diagram. The work done during a cycle isView Solution
- 10A gas at $NTP$ is suddenly compressed to one-fourth of its original volume. If $\gamma $ is supposed to be $\frac{3}{2}$, then the final pressure is........ atmosphereView Solution
